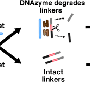Pour, shake and stir: How gold particles, DNA and water have potential to shape future of medicine
A diagnostic "cocktail" containing a single drop of blood, a dribble of water, and a dose of DNA powder with gold particles could mean rapid diagnosis and treatment of the world's leading diseases in the near future. The cocktail diagnostic is a homegrown brew being developed by University of Toronto's Institute of Biomaterials and Biomedical Engineering (IBBME) PhD student Kyryl Zagorovsky and Professor Warren Chan that could change the way infectious diseases, from HPV and HIV to malaria, are diagnosed.
And it involves the same technology used in over-the-counter pregnancy tests.
"There's been a lot of emphasis in developing simple diagnostics," says IBBME Professor and Canada Research Chair in Nanobiotechnology, Warren Chan. "The question is, how do you make it simple enough, portable enough?"
The recent winner of the NSERC E.W.R. Steacie Memorial Fellowship, Professor Chan and his lab study nanoparticles: in particular, the use of gold particles in sizes so small that they are measured in the nanoscale. Chan and his group are working on custom-designing nanoparticles to target and illuminate cancer cells and tumours, with the potential of one day being able to deliver drugs to cancer cells.
But it's a study recently published in Angewandte Chemie that's raising some interesting questions about the future of this relatively new frontier of science.
Zagorovsky's rapid diagnostic biosensor will allow technicians to test for multiple diseases at one time with one small sample, and with high accuracy and sensitivity. The biosensor relies upon gold particles in much the same vein as your average pregnancy test. With a pregnancy test, gold particles turn the test window red because the particles are linked with an antigen that detects a certain hormone in the urine of a pregnant woman.
"Gold is the best medium," explains Chan, "because it's easy to see. It emits a very intense colour."
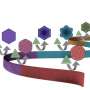
Currently scientists can target the particular disease they are searching for by linking gold particles with DNA strands: when a sample containing the disease gene (ie. Malaria) is present, it clumps the gold particles, turning the sample blue. Rather than clumping the particles together, Zagorovsky immerses the gold particles in a DNA-based enzyme solution (DNA-zyme) that, when the disease gene is introduced, 'snip' the DNA from the gold particles, turning the sample red.
"It's like a pair of scissors," Zagorovsky explains, "and the target gene activates the scissors that cut the DNA links holding gold particles together."
The advantage is that far less of the gene needs to be present for the solution to show noticeable colour changes, amplifying detection. A single DNA-zyme can clip up to 600 "links" between the target genes.
Just a single drop from a biological sample such as saliva or blood can potentially be tested in parallel, so that multiple diseases can be tested for in one sitting.
But the team has also demonstrated that they are able to transform the testing solution into a powder, making it light and far easier to ship than solutions, which degrade over time. Powder can be stored for years at a time, and offers hope that the technology can be developed into efficient, cheap, over-the-counter tests for diseases such as HIV and malaria for developing countries, where access to portable diagnostics is a necessity.
"We've now put all the pieces together," says Chan.
Journal information: Angewandte Chemie
Provided by University of Toronto Faculty of Applied Science & Engineering