Research may offer big benefits for biofuels and battling infections
(Phys.org)—Researchers at the University of Virginia School of Medicine have deciphered the secrets of the production of cellulose, the most common natural polymer on Earth, in a discovery that could have major ramifications for both biofuel production and the battle against bacterial infections.
The findings are of particular interest to the federal Department of Energy, which is seeking ways to break down plant cells more easily to facilitate the production of biofuels. Understanding the production and deposition of cellulose, the primary component of plants' cell walls, may lead to new ways to tear it down or create plants with weaker walls.
Similarly, the U.Va. findings may offer new targets for battling bacteria and preventing the spread of infections. Cellulose is one of the components that bacteria produce to create strong, spongy coatings – called "biofilms" – that allow them to clump together and cling to surfaces. The plaque that forms on teeth, for example, is a biofilm.
"If we can prevent biofilm formation, we would expect to make it easier to get rid of the bacteria – to actually kill it," U.Va. researcher Jochen Zimmer said. "And you could also prevent them from adhering to the surgical devices and other tools used in hospitals."
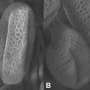
In a paper published Dec. 9 in the online edition of the journal Nature, the U.Va. researchers map out the three-dimensional architecture of the enzyme complex responsible for cellulose production. The researchers first determined the components necessary to produce and secrete cellulose and then solved the structure of the enzyme complex. Their study reveals how new cellulose polymers are extruded from a cell through a channel, a bit like a spider spinning a thread of spider silk, and how this process is intimately linked to the formation of cellulose.
Until now, the end result was understood, but the process itself was largely unknown.
The enzyme is unique in that it both produces cellulose polymers (by attaching glucose molecules) and pushes them outside the cell simultaneously; usually the division of labor is different, with production and movement either handled separately or handled by different enzymes.
"By capturing the crystal structure of part of a protein complex that both synthesizes and transfers cellulose out of a bacterium one sugar unit at a time, this work provides a window into the details of a unique cellular mechanism," says Pamela Marino of the National Institutes of Health's National Institute of General Medical Sciences, which partly funded the work. "A similar process is likely at work in the synthesis and secretion of key carbohydrate polymers in other organisms, such as hyaluronan in mammals."
In building a three-dimensional model of the atomic architecture, the U.Va. team members were surprised to observe what they had thought almost impossible: They had captured an image of a new cellulose polymer being synthesized and transported from the inside of a cell to the outside. This was most unexpected, both because the process is transitory and because the submicroscopic imaging required – a combination of X-ray diffraction and advanced math – can work only with an extremely stable and uniform ensemble of proteins.
Zimmer expects U.Va.'s findings to be significant both to biofuel production and the field of medicine, but its impact could reach even farther. He says the U.Va. team plans to extend its research to look at the biosynthesis of chitin, an essential component of the shells of insects. Preventing the formation of chitin, he says, could make for a very effective form of pest control.
The U.Va. paper, "Crystallographic snapshot of cellulose synthesis and membrane translocation," written by Jacob L.W. Morgan, Joanna Strumillo and Zimmer, was published online by Nature and will appear in a forthcoming print edition. It is the second article U.Va.'s Department of Molecular Physiology and Biological Physics has had published in Nature since Nov. 15.
More information: www.nature.com/nature/journal/ … ull/nature11744.html
Journal information: Nature
Provided by University of Virginia