Genome-wide methylation map of disease-causing E. coli reveals surprises
(Phys.org)—A new DNA sequencing technique has enabled researchers to map for the first time the influential chemical modifications known as methylation marks throughout the genome of a pathogenic bacterium. By comparing these patterns between related strains of the bacteria, they stumbled upon a way that viruses that infect bacteria (known as bacteriophages) can dramatically alter their host.
Howard Hughes Medical Institute investigator Matthew K. Waldor of Brigham and Women's Hospital, led the new study in collaboration with Eric Schadt at Mount Sinai School of Medicine. Their findings were published November 8, 2012, in the journal Nature Biotechnology.
Waldor had been studying the strain of E. coli blamed for the large 2011 outbreak of illness in Germany. He says it was clear from the early stages of the outbreak that the pathogen causing the illness were not typical, and he was curious about what gave rise to their unusual virulence. In the course of their investigation, he and his colleagues observed that certain genes were methylated differently in the disease-causing E. coli strain (E. coli O104:H4) than they were in less virulent strains.
An organism's essential genetic blueprint lies in the sequence of nucleotides that make up its DNA, but additional information is encoded in chemical modifications to those nucleotides. In animals and plants, methylation—the addition of methyl groups to specific DNA sites—is known to turn off genes. In a few model bacterial species, DNA modification is known to influence chromosome replication, gene expression, and virulence. But scientists lack a complete picture of the effects of DNA methylation in bacterial genomes.
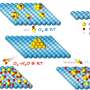
When Waldor and his colleagues investigated the altered pattern of methylation they had observed, they noticed that a unique bacteriophage (a virus) had infected the virulent E. coli strain. Furthermore, when that bacteriophage invaded the bacterial cell, it came equipped with a protein that can add methyl groups to their DNA.
"We wondered whether the phage's methylation system would influence the methylation of the bacteria it infected," says Waldor, "and whether it could even influence the virulence of the organism."
To answer this question, Waldor's team turned to a relatively new technique called single-molecule real-time (SMRT) DNA sequencing. Most methods for sequencing DNA report only the sequence of adenines, cytosines, guanines, and thymines – the four nucleotides, or bases, that make up the genetic code. But SMRT sequencing works differently. "With this technique, you monitor DNA synthesis and at the same time you get information about the order of the bases, you also get information about the kinetics of how each base is added," Waldor explains. In 2010, researchers at Pacific Biosciences discovered that chemical modifications of the bases change these kinetics—the addition of a base might be slowed down if the template base has a methyl group attached, for example.
That suggested that the chemical modifications of genes could be mapped out using SMRT sequencing. Waldor's group went even further than analyzing a single gene: they used SMRT to map the methylation patterns of the entire E.coli O104:H4 genome. They found more than 50,000 methylated sites. "Our paper is the first to show that this technique really can be used on a genome-wide level with single nucleotide resolution," says Waldor.
The scientists went on to show that the E. coli strain they were studying has eleven enzymes for controlling methylation. Seven of these enzymes, called methyltransferases, had never been researched before. Waldor's group determined what gene sequences these methyltransferases tended to add methyl groups to. Then they devoted their attention to the methyltransferase donated to the disease-causing E.coli O104:H4 from the bacteriophage that had infected it.
When E. coli O104:H4 became infected with the phage, not only were thousands of methylation sites added, but more than a third of the bacteria's genes had altered patterns of expression. "It was startling to see that infection with this virus would have such a profound global effect on transcription throughout the genome," says Waldor. The presence of the viral methyltransferase influenced the growth rate of the bacteria, though not ultimately its overall virulence.
Further work on which methyl groups are key to changing E. coli's properties will help researchers understand the role of methylation in the bacteria's life cycles, infectivity, and perhaps even drug resistance, says Waldor. And the SMRT technique can now be applied to other strains and types of bacteria. For now, the method only works in bacteria, but Waldor expects that to change quickly.
"This is like having a new microscope that can see things never before visible," he says. "It will enable a new generation of studies on the significance of DNA modification in the bacterial kingdom and, not too far off we think, in all kingdoms of life."
More information: www.hhmi.org/research/investigators/waldor.html
Journal information: Nature Biotechnology
Provided by Howard Hughes Medical Institute