Innate immunity of plants against viruses does not act as anticipated
The so-called immune receptor Rx, conferring the resistance of some potato cultivars against potato virus X, does recognise the viruses in the cytoplasm, but – unexpectedly – its presence in the nucleus is also required for an effective defence response. Erik Slootweg and Wladimir Tameling, scientists of two different Chair Groups of Wageningen University, The Netherlands, have with a number of colleagues published this striking finding in two papers published in the same issue of the scientific journal The Plant Cell.
The finding is important because other plants and even animals and humans have a similar innate immune system effective against pathogens. This discovery in plants might in the long term lead to the development of crops with a defence mechanism that is very hard to be broken by viruses.
Potato plants can defend themselves against Potato virus X (PVX) when they contain the resistance gene coding for the Rx protein. This gene belongs to a large group of genes coding for immune receptors, proteins involved in innate immunity against pathogens. Such immune receptors are found in plants, animals and humans. A virus penetrating a cell and entering the cytoplasm is recognised by the immune receptor as intruder, followed by the initiation of a rapid defence reaction.
Rx is a large protein. This means that it cannot easily move from the cytoplasm, where it is made, to the nucleus. Such large proteins that are nevertheless required in the nucleus generally have a characteristic part, the so-called nuclear localisation signal. The cell machinery uses this part of the protein as a sort of ‘postal code’ to target and transport the protein into the nucleus. But the Rx protein does not contain such a postal code. The immune receptor was therefore expected to be only present and active in the cytoplasm.
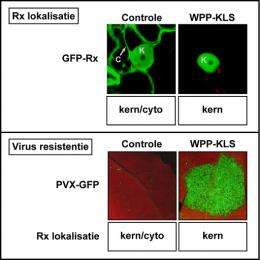
To unravel the mode of action of the Rx immune receptor, the Wageningen scientists attached a green fluorescent protein to the Rx protein. Using a fluorescence microscope, this enabled them to establish the location of the Rx protein in living plant cells. This is how they discovered that the immune receptor was not only present in the cytoplasm but indeed also in the nucleus.
Because Rx does not contain a ‘postal code’ for transport to the nucleus it will probably be transported into the nucleus in a unique way. The initial view was that the protein RanGAP2, which is binding to Rx, would stimulate this transport. But RanGAP2 was found to perform an exactly opposite role, viz. it retained part of the Rx molecules in the cytoplasm.
The Wageningen scientists wanted to know whether the presence of Rx in the nucleus is also playing a role in the actual defence against viruses. In experiments they prevented the immune receptor molecules from getting into the nucleus or – conversely - they forced all these molecules to be transported from the cytoplasm into the nucleus. In both cases the plants were found to be unable to defend themselves against the virus. A certain balance between the amount of Rx in nucleus and cytoplasm apparently is crucial for the initiation of an effective defence response. Because RanGAP2 retains part of the Rx molecules in the cytoplasm, RanGAP2 gives the plant the right balance between the amount of Rx in the cell nuclei and the amount of Rx in the cytoplasm. This means that the role of RanGAP2 is crucial in the defence against pathogens.
Similar immune receptors are found in plants, animals and humans, and provide innate immunity against pathogens, including viruses. This makes the finding of the Wageningen scientists also relevant for a better understanding of our own innate immune system. The discovery may in the long term lead to the development of plants with a defence against viruses which would be difficult for them to circumvent.
Provided by Wageningen University