Hybrid Nanoparticles Image and Treat Tumors
(PhysOrg.com) -- By combining a magnetic nanoparticle, a fluorescent quantum dot, and an anticancer drug within a lipid-based nanoparticle, a multi-institutional research team headed by members of the National Cancer Institute’s (NCI) Alliance for Nanotechnology in Cancer has created a single agent that can image and treat tumors. In addition, this new nanoparticle is able to avoid detection by the immune system, enabling the particle to remain in the body for extended periods of time.
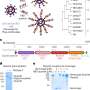
“The idea involves encapsulating imaging agents and drugs into a protective ‘mothership’ that evades the natural processes that normally would remove these payloads if they were unprotected,” said Michael Sailor, Ph.D., an Alliance member at the University of California, San Diego, who led this research effort. Other Alliance members who participated in this study include Sangeeta Bhatia, M.D., Ph.D., Massachusetts Institute of Technology, and Erkki Ruoslahti, M.D., Ph.D., Burnham Institute for Medical Research at the University of California, Santa Barbara. The researchers published the results of their work in the journal Angewandte Chemie International Edition.
“Many drugs look promising in the laboratory but fail in humans because they do not reach the diseased tissue in time or at concentrations high enough to be effective,” added Dr. Bhatia. “These drugs don’t have the capability to avoid the body’s natural defenses or to discriminate their intended targets from healthy tissues. In addition, we lack the tools to detect diseases such as cancer at the earliest stages of development, when therapies can be most effective.”
The researchers designed the hull of their motherships to evade detection by constructing them of lipids modified with poly(ethylene glycol) (PEG). The researchers also designed the material of the hull to be strong enough to prevent accidental release of the mothership’s cargo while circulating through the bloodstream. Tethered to the surface of the hull is a protein called F3, a molecule that sticks to cancer cells. Prepared in Dr. Ruoslahti’s laboratory, F3 was engineered to specifically home in on tumor cell surfaces and then transport itself into their nuclei.
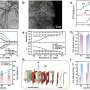
The researchers loaded their mothership nanoparticles with three payloads before injecting them in mice. Two types of nanoparticles, superparamagnetic iron oxide and fluorescent quantum dots, were placed in the ship’s cargo hold, along with the anticancer drug doxorubicin. The iron oxide nanoparticles allow the ships to show up in a magnetic resonance imaging (MRI) scan, and the quantum dots can be seen with another type of imaging tool, a fluorescence scanner.
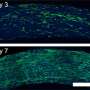
“The fluorescence image provides higher resolution than MRI,” said Dr. Sailor. “One can imagine a surgeon identifying the specific location of a tumor in the body before surgery with an MRI scan, then using fluorescence imaging to find and remove all parts of the tumor during the operation.”
To its surprise, the team found that a single mothership can carry multiple iron oxide nanoparticles, which increases their brightness in the MRI image. “The ability of these nanostructures to carry more than one superparamagnetic nanoparticle makes them easier to see by MRI, which should translate to earlier detection of smaller tumors,” said Dr. Sailor. “The fact that the ships can carry very dissimilar payloads—a magnetic nanoparticle, a fluorescent quantum dot, and a small molecule drug—was a real surprise.”
This work, which is detailed in the paper “Micellar Hybrid Nanoparticles for Simultaneous Magnetofluorescent Imaging and Drug Delivery,” was supported by the NCI Alliance for Nanotechnology in Cancer. (dx.doi.org/doi:10.1002/anie.200801810)
Provided by National Cancer Institute