New catalyst transforms carbon dioxide from industrial emissions into commonly used chemicals
A low-cost, tin-based catalyst can selectively convert carbon dioxide to three widely produced chemicals—ethanol, acetic acid and formic acid.
A low-cost, tin-based catalyst can selectively convert carbon dioxide to three widely produced chemicals—ethanol, acetic acid and formic acid.
Analytical Chemistry
14 hours ago
0
201

A costly step in the process of taking carbon dioxide emissions and converting them into useful products such as biofuels and pharmaceuticals may not be necessary, according to University of Michigan researchers.
Nanomaterials
16 hours ago
0
6
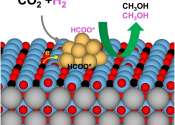
A team of scientists led by the Department of Energy's Oak Ridge National Laboratory has found an unconventional way to improve catalysts made of more than one material. The solution demonstrates a path to designing catalysts ...
Analytical Chemistry
May 16, 2024
0
279
A research team led by Wang Guozhong from the Hefei Institutes of Physical Science of the Chinese Academy of Sciences encapsulated metallic nickel in a nitrogen-doped carbon-silica composite (SiO2@Ni@NC) as a catalyst, which ...
Analytical Chemistry
May 16, 2024
0
18

Prof. Wang Qi's research group from the Hefei Institutes of Physical Science of the Chinese Academy of Sciences has synthesized iron- and nitrogen-co-doped CoFeP-N nanowires for high-efficiency electrocatalytic water splitting.
Nanomaterials
May 16, 2024
0
58

Methane, a greenhouse gas that contributes significantly to global warming, is also an important source of energy and an essential chemical resource. When used as a chemical feedstock, methane is typically converted into ...
Analytical Chemistry
May 15, 2024
0
254

A team of researchers has made significant strides in understanding metal-nitrogen-carbon (M-N-C) catalysts, offering alternatives to expensive platinum-group-metal (PGM) catalysts and a pathway to a greener future.
Analytical Chemistry
May 15, 2024
0
1

A research team has proposed new understandings of the poisoning effect of carbon deposition during carbon dioxide reduction reaction (CO2RR) on the active sites of Cu electrode. The study was published in Precision Chemistry.
Analytical Chemistry
May 14, 2024
0
1

Rapid growth in global energy demand has caused massive depletion of traditional fossil fuels and serious environmental problems, and there is no doubt that the development of efficient energy storage and conversion technologies ...
Analytical Chemistry
May 14, 2024
0
5

The quest for more productive catalysts for the creation of sustainable fuels and commodity chemicals via electrochemical reactions just became easier.
Analytical Chemistry
May 13, 2024
0
18
Catalysis is the process in which the rate of a chemical reaction is either increased or decreased by means of a chemical substance known as a catalyst. Unlike other reagents that participate in the chemical reaction, a catalyst is not consumed by the reaction itself. The catalyst may participate in multiple chemical transformations. Catalysts that speed the reaction are called positive catalysts. Catalysts that slow down the reaction are called negative catalysts or inhibitors. Substances that increase the activity of catalysts are called promoters and substances that deactivate catalysts are called catalytic poisons. For instance, in the reduction of ethyne to ethene, the catalyst is palladium (Pd) partly "poisoned" with lead(II) acetate (Pb(CH3COO)2). Without the deactivation of the catalyst, the ethene produced will be further reduced to ethane.
The general feature of catalysis is that the catalytic reaction has a lower rate-limiting free energy change to the transition state than the corresponding uncatalyzed reaction, resulting in a larger reaction rate at the same temperature. However, the mechanistic origin of catalysis is complex. Catalysts may affect the reaction environment favorably, e.g. acid catalysts for reactions of carbonyl compounds, form specific intermediates that are not produced naturally, such as osmate esters in osmium tetroxide-catalyzed dihydroxylation of alkenes, or cause lysis of reagents to reactive forms, such as atomic hydrogen in catalytic hydrogenation.
Kinetically, catalytic reactions behave like typical chemical reactions, i.e. the reaction rate depends on the frequency of contact of the reactants in the rate-determining step. Usually, the catalyst participates in this slow step, and rates are limited by amount of catalyst. In heterogeneous catalysis, the diffusion of reagents to the surface and diffusion of products from the surface can be rate determining. Analogous events associated with substrate binding and product dissociation apply to homogeneous catalysts.
Although catalysts are not consumed by the reaction itself, they may be inhibited, deactivated or destroyed by secondary processes. In heterogeneous catalysis, typical secondary processes include coking where the catalyst becomes covered by polymeric side products. Additionally, heterogeneous catalysts can dissolve into the solution in a solid-liquid system or evaporate in a solid-gas system.
This text uses material from Wikipedia, licensed under CC BY-SA