The Indian villagers who lost their homes to the sea
The gentle roar of the ocean lulled Indian mother-of-two Banita Behra to sleep each night, until one day the encroaching tide reached her doorstep.

The gentle roar of the ocean lulled Indian mother-of-two Banita Behra to sleep each night, until one day the encroaching tide reached her doorstep.
Environment
4 hours ago
0
25
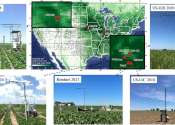
In recent years, the scientific community has increasingly turned its attention to sustainable agriculture, aiming to maximize crop yield while minimizing environmental impact. A crucial aspect of this research involves understanding ...
Ecology
15 hours ago
0
62

Researchers at the University of Bayreuth have gained new insights in the field of high-pressure carbon chemistry: They synthesized two new carbides—compounds of carbon and another chemical element—with unique structures. ...
Analytical Chemistry
19 hours ago
0
13

New University of Minnesota research suggests "leaf glow" provides vital information on vegetation dynamics in Arctic and boreal ecosystems like Minnesota's forests and wetlands, which are among the fastest warming in the ...
Plants & Animals
14 hours ago
0
13

Arctic riverbanks are typically resilient, thanks to the power of permafrost. This permanently frozen soil locks in sediment, leading to low erosion rates. But as Arctic river water warms due to climate change, some researchers ...
Earth Sciences
18 hours ago
0
12

The Scottish government's decision to row back on its 2030 climate pledge illustrates the crux of any target: it's easy to set one with a big political flourish, but harder to follow through with a careful plan to achieve ...
Environment
18 hours ago
0
17

Water erosion is the most active process controlling soil formation and evolution, which can affect the redistribution of carbon between terrestrial, aquatic, and atmospheric ecosystems. Erosion-induced organic carbon dynamic ...
Earth Sciences
14 hours ago
0
1
Carbon (pronounced /ˈkɑrbən/) is the chemical element with symbol C and atomic number 6. As a member of group 14 on the periodic table, it is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. There are three naturally occurring isotopes, with 12C and 13C being stable, while 14C is radioactive, decaying with a half-life of about 5730 years. Carbon is one of the few elements known since antiquity. The name "carbon" comes from Latin language carbo, coal, and, in some Romance and Slavic languages, the word carbon can refer both to the element and to coal.
There are several allotropes of carbon of which the best known are graphite, diamond, and amorphous carbon. The physical properties of carbon vary widely with the allotropic form. For example, diamond is highly transparent, while graphite is opaque and black. Diamond is among the hardest materials known, while graphite is soft enough to form a streak on paper (hence its name, from the Greek word "to write"). Diamond has a very low electrical conductivity, while graphite is a very good conductor. Under normal conditions, diamond has the highest thermal conductivity of all known materials. All the allotropic forms are solids under normal conditions but graphite is the most thermodynamically stable.
All forms of carbon are highly stable, requiring high temperature to react even with oxygen. The most common oxidation state of carbon in inorganic compounds is +4, while +2 is found in carbon monoxide and other transition metal carbonyl complexes. The largest sources of inorganic carbon are limestones, dolomites and carbon dioxide, but significant quantities occur in organic deposits of coal, peat, oil and methane clathrates. Carbon forms more compounds than any other element, with almost ten million pure organic compounds described to date, which in turn are a tiny fraction of such compounds that are theoretically possible under standard conditions.
Carbon is one of the least abundant elements in the Earth's crust, but the fourth most abundant element in the universe by mass after hydrogen, helium, and oxygen. It is present in all known lifeforms, and in the human body carbon is the second most abundant element by mass (about 18.5%) after oxygen. This abundance, together with the unique diversity of organic compounds and their unusual polymer-forming ability at the temperatures commonly encountered on Earth, make this element the chemical basis of all known life.
This text uses material from Wikipedia, licensed under CC BY-SA