Unraveling life's origin: Five key breakthroughs from the past five years
There is still so much we don't understand about the origin of life on Earth.

There is still so much we don't understand about the origin of life on Earth.
Cell & Microbiology
May 4, 2024
0
94

Reforestation efforts to restock depleted forests are important for addressing climate change and for both capturing and restoring carbon from the Earth's atmosphere. These types of solutions to mitigate carbon emissions ...
Environment
May 4, 2024
1
9

New research involving the University of East Anglia (UEA) suggests that countries' current plans to remove CO2 from the atmosphere will not be enough to comply with the 1.5ºC warming limit set out under the Paris Agreement.
Environment
May 3, 2024
1
51

Recent reports of NASA's James Webb Space Telescope finding signs of life on a distant planet understandably sparked excitement. A new study challenges this finding, but also outlines how the telescope might verify the presence ...
Astrobiology
May 2, 2024
0
74

Scientists at the Department of Energy's Oak Ridge National Laboratory have developed lubricant additives that protect both water turbine equipment and the surrounding environment.
Analytical Chemistry
May 2, 2024
1
95
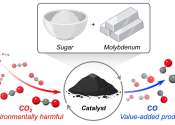
A new catalyst made from an inexpensive, abundant metal and common table sugar has the power to destroy carbon dioxide (CO2) gas.
Analytical Chemistry
May 2, 2024
0
17

A research team at the University of Pittsburgh led by Alexander Star, a chemistry professor in the Kenneth P. Dietrich School of Arts and Sciences, has developed a fentanyl sensor that is six orders of magnitude more sensitive ...
Bio & Medicine
May 2, 2024
0
90

Some of the ocean's tiniest organisms get swept into underwater currents that act as a conduit that shuttles them from the sunny surface to deeper, darker depths where they play a huge role in affecting the ocean's chemistry ...
Ecology
May 2, 2024
0
47

In early spring, George Okoko was perched on a ledge 15 feet up a crumbly cliff, trying to whack off a basketball-size piece of rock with a hammer and chisel. The locale was suburban Berkeley Heights, N.J. The rock was basalt, ...
Earth Sciences
May 2, 2024
1
1

The Environmental Protection Agency (EPA) is underestimating methane emissions from landfills, urban areas and U.S. states, according to a new study led by researchers at the Harvard John A. Paulson School of Engineering ...
Environment
May 1, 2024
0
45
Carbon (pronounced /ˈkɑrbən/) is the chemical element with symbol C and atomic number 6. As a member of group 14 on the periodic table, it is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. There are three naturally occurring isotopes, with 12C and 13C being stable, while 14C is radioactive, decaying with a half-life of about 5730 years. Carbon is one of the few elements known since antiquity. The name "carbon" comes from Latin language carbo, coal, and, in some Romance and Slavic languages, the word carbon can refer both to the element and to coal.
There are several allotropes of carbon of which the best known are graphite, diamond, and amorphous carbon. The physical properties of carbon vary widely with the allotropic form. For example, diamond is highly transparent, while graphite is opaque and black. Diamond is among the hardest materials known, while graphite is soft enough to form a streak on paper (hence its name, from the Greek word "to write"). Diamond has a very low electrical conductivity, while graphite is a very good conductor. Under normal conditions, diamond has the highest thermal conductivity of all known materials. All the allotropic forms are solids under normal conditions but graphite is the most thermodynamically stable.
All forms of carbon are highly stable, requiring high temperature to react even with oxygen. The most common oxidation state of carbon in inorganic compounds is +4, while +2 is found in carbon monoxide and other transition metal carbonyl complexes. The largest sources of inorganic carbon are limestones, dolomites and carbon dioxide, but significant quantities occur in organic deposits of coal, peat, oil and methane clathrates. Carbon forms more compounds than any other element, with almost ten million pure organic compounds described to date, which in turn are a tiny fraction of such compounds that are theoretically possible under standard conditions.
Carbon is one of the least abundant elements in the Earth's crust, but the fourth most abundant element in the universe by mass after hydrogen, helium, and oxygen. It is present in all known lifeforms, and in the human body carbon is the second most abundant element by mass (about 18.5%) after oxygen. This abundance, together with the unique diversity of organic compounds and their unusual polymer-forming ability at the temperatures commonly encountered on Earth, make this element the chemical basis of all known life.
This text uses material from Wikipedia, licensed under CC BY-SA