This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
A new reaction to enhance aromatic ketone use in chemical synthesis
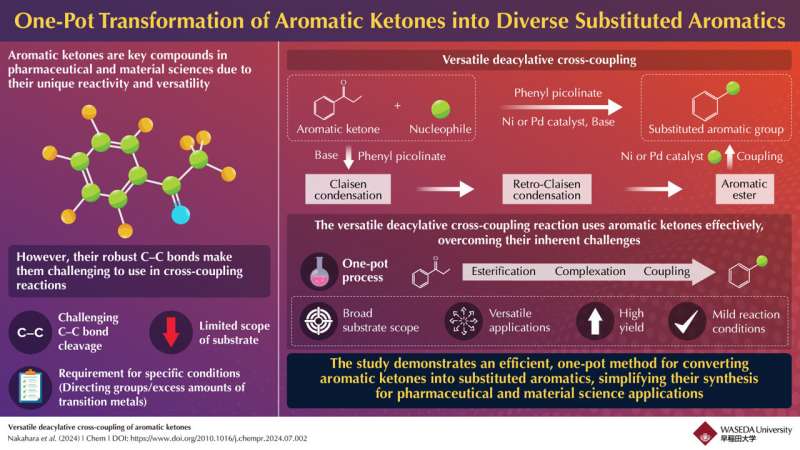
Aromatic ketones have long been valuable intermediates in chemical synthesis, particularly in cross-coupling reactions where different chemical entities are combined to form new compounds. For instance, a process called deacylative cross-coupling removes the acyl group from the aromatic ketone, allowing it to bond with other chemicals and produce a wide variety of useful compounds. These reactions are crucial for producing a wide array of aromatic compounds used in various industries like agrochemicals.
However, the utility of aromatic ketones has been limited due to the difficulty in breaking their strong carbon-carbon bonds. These robust bonds are challenging to cleave, often requiring specific conditions or catalysts.
Traditional methods for overcoming this challenge have involved complex and costly procedures, which include the use of directing groups and large amounts of transition metals. These limitations can complicate the process and increase overall costs, hindering the broader application of aromatic ketones in various industrial and synthetic contexts.
To address traditional challenges in using aromatic ketones, a team of researchers led by Professor Junichiro Yamaguchi from the Faculty of Science and Engineering, School of Advanced Science and Engineering at Waseda University has developed a groundbreaking one-pot method that optimizes the conversion of aromatic ketones into aromatic esters. The research, published in Chem, highlights its potential for broader use in synthetic chemistry.
This new approach significantly simplifies the reaction process by integrating a Claisen reaction and a retro-Claisen reaction into a single step. The Claisen reaction combines two molecules to form a larger intermediate compound, while the retro-Claisen reaction modifies this intermediate to produce the desired ester. By combining these steps into a one-pot process, the researchers have managed to simplify the synthesis, reduce reaction times, and minimize the need for additional purification steps.
"This advancement enhances the versatility and utility of aromatic ketones, facilitating their use in pharmaceutical synthesis and materials science," remarks Dr. Yamaguchi.
The flexibility of the new reaction method allows it to work with a wide variety of reactants, including alcohols, phenols, amines, and thiols. This technique is particularly notable because it enables seven different types of chemical transformations, turning compounds into thioethers, aryl groups, hydrogen, α-aryls, methylphosphonyl groups, amines, and ethers. This broad range of capabilities makes the method highly adaptable and useful for various chemical processes, improving how aromatic ketones can be used across different industries.
Furthermore, the catalyst system employed in this reaction has shown notable stability and reusability, making the process scalable for industrial applications.
"This reaction represents the first successful example of directly catalyzing aromatic ketones without the use of directing groups, setting the stage for future advancements in synthetic chemistry," says Dr. Yamaguchi.
More information: Hikaru Nakahara et al, Versatile deacylative cross-coupling of aromatic ketones, Chem (2024). DOI: 10.1016/j.chempr.2024.07.002
Journal information: Chem
Provided by Waseda University