This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Unraveling the behavior of nanoconfined water and ice in extreme conditions
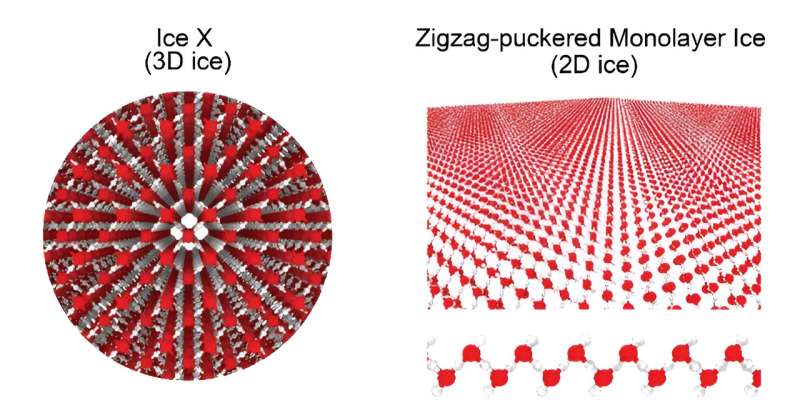
Understanding water behavior in nanopores is crucial for both science and practical applications. Scientists from City University of Hong Kong (CityU) have revealed the remarkable behavior of water and ice under high pressure and temperature, and strong confinement.
The study, titled "Rich Proton Dynamics and Phase Behaviors of Nanoconfined Ices", was published in Nature Physics.
These findings, which defy the normal behavior observed in daily life, hold immense potential for advancing our understanding of water's unusual properties in extreme environments, such as in the core of distant ice planets. The implications of this major scientific advancement span various fields, including planetary science, energy science, and nanofluidic engineering.
Led by Professor Zeng Xiaocheng, Head and Chair Professor in the Department of Materials Science and Engineering at CityU, the research team employed state-of-the-art computational methods to simulate the properties of water and ice under extreme conditions.
Through machine learning potential, crystal structure searches, path-integral molecular dynamics, and metadynamics, they conducted comprehensive simulations of mono- and bi-layer water under nanoconfinement. These simulations unveiled a range of intriguing phenomena, including two-dimensional (2D) ice-to-water melting, novel ice behavior, water splitting, and proton dynamics in nano ice.
The research team discovered 10 new 2D ice states, each exhibiting unique characteristics. Notably, they identified 2D molecular ice with a symmetric O-H-O configuration, reminiscent of the highest density 3D Ice X found on Earth. Additionally, they observed dynamic, partially ionic ice and several superionic ices. Surprisingly, these 2D ice states could be produced at much lower pressures than their 3D counterparts with similar water density, making them more accessible in laboratory conditions.
Professor Zeng emphasized the significance of these findings, stating that they represent a new frontier in understanding the physics and chemistry of water and ice under extreme conditions, particularly in the core of ice giant planets.
"The potential to create these unique ice- and water-splitting states in the laboratory, including dynamic, partially ionic and superionic ices at lower pressure than previously thought possible, is particularly exciting," said Professor Zeng.
Exploring the behavior of water and ice under different conditions, especially when nanoconfinement is considered, is a profoundly complex task.
The research team tackled this challenge through an extensive number of simulations of molecular dynamics and path-integral molecular dynamics, generating a vast dataset. Extracting meaningful insights from this enormous amount of data posed a significant data analysis challenge, requiring meticulous exploration.
These findings pave the way for future research into the mysteries of ice giant planets and the fundamental properties of water. The next phase of this research involves experimental validation of the computational predictions and exploration of practical applications.
Professor Zeng expressed enthusiasm about the potential of this research to deepen our understanding of water, ice, and water-splitting in extreme environments, while also opening new frontiers in nanoscience and planetary research.
More information: Jian Jiang et al, Rich proton dynamics and phase behaviours of nanoconfined ices, Nature Physics (2024). DOI: 10.1038/s41567-023-02341-8
Journal information: Nature Physics
Provided by City University of Hong Kong





















