This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers reveal mechanism behind most common mammalian mRNA modification
RNA—in the form of messenger RNA (mRNA), ribosomal RNA (rRNA), and transfer RNA (tRNA)—transforms the genome coded by DNA into proteins that form the backbone of all cellular functions. However, biochemical modifications to RNA frequently occur, with a subsequent influence on gene expression and the potential to cause disease. The sum of these functionally relevant changes to RNA is called the epitranscriptome.
The most common mammalian mRNA modification currently known is the methylation of nitrogen at position 6 in the adenosine base of mRNA. This modification is known as N6-methyladenosine (m6A), which is present in 0.2%–0.6% of all adenosines. Nevertheless, how this common methylation occurs and is controlled has not been well understood.
Now, however, a new study led by Prof. Ren Jie and Prof. Yang Yungui from the Beijing Institute of Genomics of the Chinese Academy of Sciences (China National Center for Bioinformation) has identified the mechanism underlying the m6A RNA modification. The study was published in Molecular Cell on April 2.
The m6A modification affects the entire life cycle of mRNA, including pre-mRNA splicing, 3′-end processing, nuclear export, translation, stability, and decay. It is catalyzed by the methyltransferase complex (MTC). This complex, in conjunction with the inhibitory role of the exon junction complex, collectively shapes the distribution profile of m6A, which spreads over the gene body and exhibits specific enrichment toward stop codons.
This pattern is established co-transcriptionally—i.e., before RNA synthesis is complete and while the nascent RNA is still tethered to the DNA by the polymerase—and is maintained in a steady state. However, how MTC recruitment works has been unclear.
In previous work, the researchers had identified m6A on R-loops—three-stranded nucleic acid structures consisting of a DNA:RNA hybrid and a displaced strand of DNA. R-loops play a vital role in regulating gene expression, DNA replication, and DNA and histone modification.
Based on the assumption that the majority of R-loops are formed co-transcriptionally, the researchers hypothesized that R-loops may play a role in the co-transcriptional modification of m6A.
To test this hypothesis and investigate how m6A might be installed co-transcriptionally, the researchers performed a protein co-immunoprecipitation (co-IP) assay in combination with mass spectrometry, using the MTC subunits METTL3, METTL14, and WTAP as "bait."
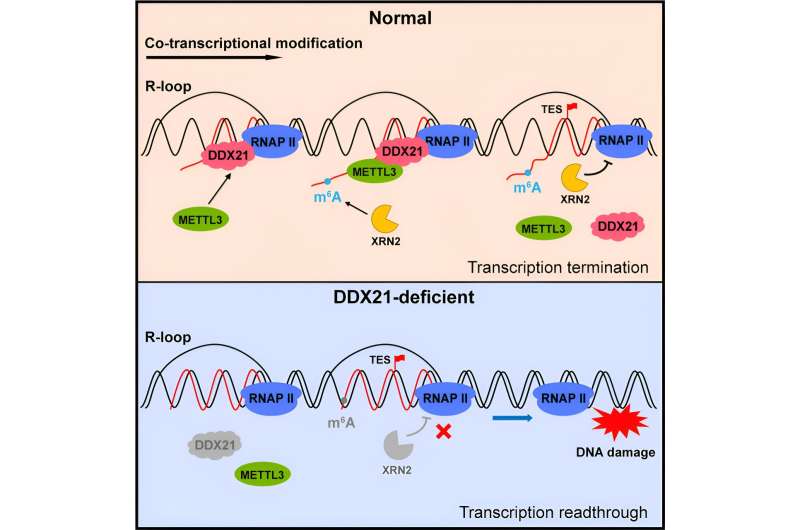
In this process, the researchers observed that R-loops, METTL3, and the helicase DDX21 strongly colocalize. (A helicase is an enzyme that separates the two strands of a helical nucleic acid structure.) They discovered that R-loops provide anchor points for recruiting DDX21 to chromatin-bound nascent transcripts, and that DDX21, in turn, recruits METTL3 to its substrate.
They noted that depletion of R-loops or DDX21 leads to a notable decrease in global m6A levels, particularly at gene ends. They thus concluded that DDX21, in collaboration with R-loops and METTL3, guides the interplay of co-transcriptional RNA modification.
Furthermore, the researchers revealed that DDX21, in conjunction with METTL3, the resulting m6A modification, and its reader YTHDC1, plays a crucial role in facilitating XRN2-mediated transcription termination, thereby ensuring genome stability. Disruption of these concerted actions at any step may lead to incomplete transcription termination or readthrough, potentially resulting in DNA damage.
This study uncovered new functions of DDX21 in promoting co-transcriptional m6A deposition, thus identifying the missing link between co-transcriptional methylation and its regulatory roles in coordinating transcription termination and maintaining genome stability.
Further exploration of the DDX21–METTL3–m6A axis may contribute to innovative strategies for addressing diseases associated with dysregulated m6A metabolism, including cancer and neurological disorders.
More information: Jin-Dong Hao et al, DDX21 mediates co-transcriptional RNA m6A modification to promote transcription termination and genome stability, Molecular Cell (2024). DOI: 10.1016/j.molcel.2024.03.006
Journal information: Molecular Cell
Provided by Chinese Academy of Sciences