This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Our ligaments and bones don't grow the way we thought, new research finds
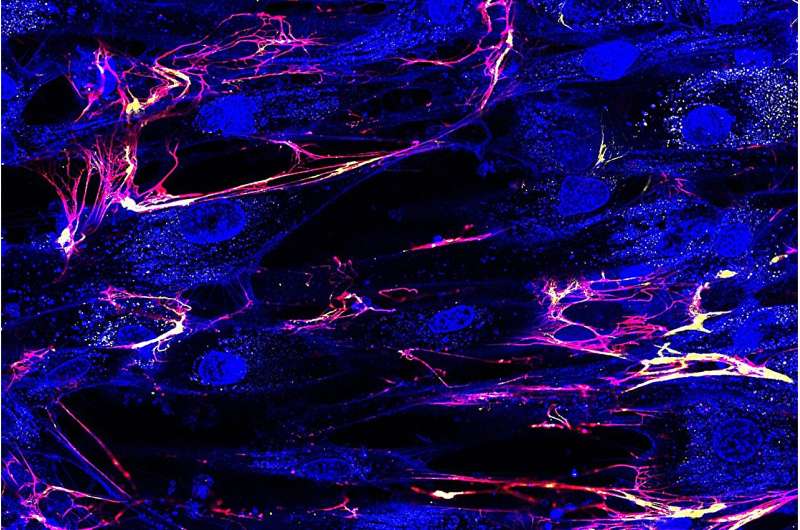
New research by Northeastern scientists questions the long-held belief that the connective tissues that give us mechanical strength, such as tendons, ligaments, bones and skin, form in the human body by cells coming together.
Instead, our tissues are more likely formed by cells pulling apart, according to the research published today in Matter.
It's long been held that human cells form and connect tissues together using patterns encoded into our DNA, says Jeff Ruberti, professor of bioengineering at Northeastern University and a co-author of the study.
"We don't really understand how tiny cells working together with molecules much smaller than themselves are able to integrate and establish these lovely, mechanically efficient patterns that become us over time," he says. "The theory has long been that the cells basically are individually depositing, or printing the tissues, out using a pattern or algorithm."
But that's just a theory, he explains. Ruberti and three researchers who worked in his lab have now challenged that, providing evidence that collagen, the main protein that forms much of our tissue, is not pre-fabricated and positioned by the cells, but rather formed by our cells cooperatively pulling apart from each other.
"This material tends to accumulate in the path of force, where it reaches a lower energy state but is under high tensile load. So, what we propose is that the cells and your body work together to produce structure by literally creating lines of tension along which collagen precipitates into structure. In short, the force causes the structure that then resists the force that caused it."
Ruberti conducted the research with Seyed Mohammad Siadat, a research scientist in his lab, Alexandra Silverman, who graduated from Northeastern with a master's in bioengineering and Jason Olszewski, a fourth-year bioengineering major.
For the study, the team, using donated human cells, created a model of a human cornea, using high-grade microscopes and other measuring and diagnostic tools to observe the biomechanical processes that occur as the cornea was formed. The fact that the process could be seen in real time was key as previous studies in this domain largely relied on still images.
"We took extracted cells out from a donor eye, we put them on a dish, and as they worked to reform tissue, we observed that process," Ruberti says.
What they observed was that cells were pulling apart to form the structure, identifying five unique types of pulls.
Ruberti's lab first proposed this phenomena in 2016, after former Northeastern graduate student Jeff Paten performed experiments that showed for the first time if you pulled on collagen monomers, they'd crystallize into structures. Ruberti predicted that cells could do the same.
The research will be used to help inform therapeutic processes at the Northeastern spinout company BrillantStrings Therapeutics, which he and Paten have founded. The discovery could help lead to better treatments for fibrosis and other medical conditions that cause wounds to heal poorly, Ruberti says.
"It opens up myriad of possibilities, from sculpting tissues by judiciously applying loads to monomers to create structures to treat fibrosis where this process has gone wrong," he adds.
"Jeff's theory initially seems a little counterintuitive because you would think the cells have a nucleus and that's kind of the brain of the cells," says Silverman. "Of course they should know where the collagen would go and put it in that path to create the structure. It turns everything on its head and makes you question everything you might have thought."
In discussing the findings, Ruberti gets a bit philosophical, noting that in a lot of ways animals and humans could be literally pulled into existence.
"The pulling is required for you to even form properly," he says. "The tension is critical."
More information: Alexandra A. Silverman et al, Tension in the ranks: Cooperative cell contractions drive force-dependent collagen assembly in human fibroblast culture, Matter (2024). DOI: 10.1016/j.matt.2024.01.023
Provided by Northeastern University
This story is republished courtesy of Northeastern Global News news.northeastern.edu.