February 8, 2024 feature
This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Beyond cells: Unveiling the potential of genetic circuits on single DNA molecules
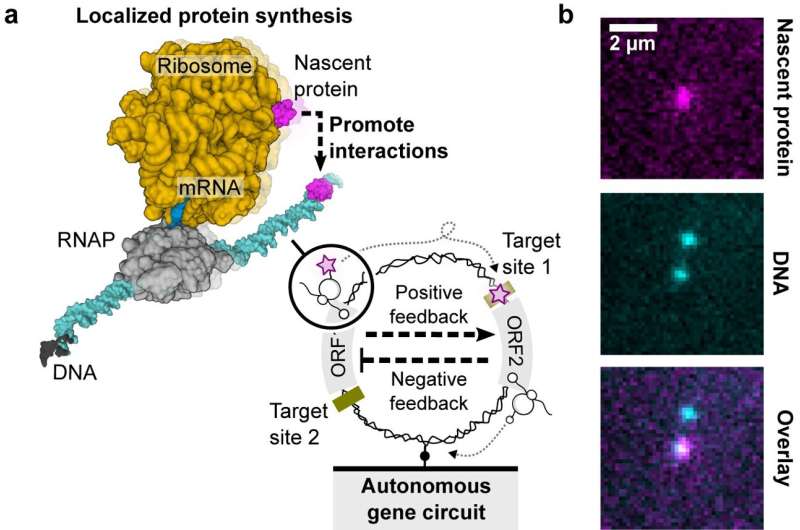
In a new Nature Communications study, researchers have explored the construction of genetic circuits on single DNA molecules, demonstrating localized protein synthesis as a guiding principle for dissipative nanodevices, offering insights into artificial cell design and nanobiotechnology applications.
The term "genetic circuit" is a metaphorical description of the complex network of genetic elements (such as genes, promoters, and regulatory proteins) within a cell that interact to control gene expression and cellular functions.
In the realm of artificial cell design, scientists aim to replicate and engineer these genetic circuits to create functional, self-contained units. These circuits act as the molecular machinery responsible for orchestrating cellular processes by precisely regulating the production of proteins and other molecules.
By understanding and manipulating these circuits, researchers can engineer artificial cells with programmable behaviors, mimicking the functionalities of natural cells.
In the context of the mentioned study, the focus is on constructing genetic circuits on single DNA molecules. This represents a novel approach as it moves away from the traditional cellular context and explores the possibility of creating genetic circuits in cell-free conditions.
First author Dr. Ferdinand Greiss from the Weizmann Institute of Science in Israel explained the researchers' motivation to Phys.org: "We are trying to reconstitute biological processes outside the complex circuitry of living cells, hopefully improving our understanding of nature's guiding principles. The research is directed toward the construction of future artificial cells, and single DNA molecules could be the genetic foundation for such."
Gene regulation
Gene regulation is the process by which cells control the expression of genes, determining when and to what extent a gene's information is utilized in the synthesis of functional molecules like proteins or RNA. It plays a crucial role in maintaining cellular functions, responding to environmental changes, and ensuring proper development.
The regulation of gene expression involves transcription and translation. During transcription, a specific segment of DNA serves as a template for the synthesis of complementary mRNA molecules by RNA polymerase. This mRNA carries the genetic code from the nucleus to the cytoplasm, where translation occurs.
Translation involves the conversion of mRNA into proteins. Ribosomes read the mRNA sequence, facilitating the assembly of amino acids into a polypeptide chain, forming the protein encoded by the gene.
"In prokaryotic systems, the transcription and translation processes are coupled. This means that once the RNA polymerase produces mRNA from DNA, the ribosome can find the ribosomal binding site on the nascent mRNA to start synthesizing the protein. The nascent protein can fold and function while still tethered to the DNA by the RNA polymerase-mRNA-ribosome complex. After termination of either transcription or translation, the nascent protein falls off the DNA and disperses into the bulk solution," explained co-author Dr. Shirley Shulman Daube from the Weizmann Institute of Science in Israel.
The significance lies in the increased local concentration of nascent proteins, which is about 1,000 times higher than the surrounding bulk solution. This spatial organization and concentration boost could have implications for cellular functions and potentially play a role in the construction of artificial cells using single DNA molecules.
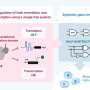
Building a genetic circuit on a single DNA molecule
"Genetic circuits are based on genetically encoded molecules, such as transcription factors, that are produced from DNA and bind back to the DNA to regulate their own and other molecules' production," said co-author Dr. Vincent Noireaux from the University of Minnesota.
To build the genetic circuit on a single DNA molecule, the researchers designed specific sequences with lambda bacteriophage (E. coli) genes.
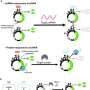
The genetic circuit involved a negative cascade, guided by the CI repressor gene and its operator binding site, intricately controlling the HT gene. This HT gene encoded the HaloTag (HT) protein, a crucial element for visualizing nascent proteins on individual DNA molecules.
The study implemented stringent conditions, including low DNA surface density, to ensure precise localized protein synthesis.
Simultaneously, a positive cascade unfolded with the fusion of the T7 bacteriophage RNA polymerase (HT-T7 RNAP) and the HT protein, enabling real-time monitoring of gene expression through a downstream reporter gene, GFP.
A far-red fluorogenic dye (MaP655-Halo) enhanced the detection of nascent proteins, providing a comprehensive view of the genetic circuit's dynamics.
The negative cascade, or suppression, regulates and inhibits the production of specific proteins under certain conditions. On the other hand, positive cascades contribute to the activation and expression of specific genes within the genetic circuit.
The research went beyond mere observation, incorporating a feedback circuit featuring a synthetic dCro repressor. This component was crucial in regulating gene expression through a meticulously designed synthetic promoter.
Free of cellular confinement
The researchers discovered that localized protein synthesis on a single DNA molecule can drive genetic circuits in cell-free conditions without the confinement of cellular compartments. The dynamics of genetic circuits were meticulously observed in very dilute conditions.
Lead author Dr. Roy Bar-Ziv from the Weizmann Institute of Science in Israel highlighted the significance of their findings: "The regulation of gene expression depends on proteins binding to the DNA, blocking or increasing the activity of a gene. The binding requires high concentrations of proteins to find and bind specific sequences on the DNA molecule. Unexpectedly, we find that localized protein synthesis can transiently increase the concentration long enough for proteins to do the same without cellular confinement."
In essence, the finding challenges the conventional notion that high concentrations are essential for gene regulation, introducing a novel aspect of localized protein synthesis as a means to influence genetic circuits in cell-free conditions.
For future work, the researchers envision leveraging localized protein synthesis as a guiding principle to enhance the functionality of artificial cells constructed from single DNA molecules, addressing challenges at low concentrations. They also foresee potential applications in self-encoded nanodevices and plan to explore correlations between DNA structure, gene expression dynamics, and protein synthesis.
The research also involved contributions from Nicolas Lardon with Prof. Kai Johnsson at the MPI for Medical Research, who developed the fluorogenic dye (MaP655-Halo); Yoav Barak, who helped with optimizing the DNA preparation; and Leonie Schütz with Prof. Elmar Weinhold, who pioneered the development of methyltransferases for site-specific DNA modifications with biotins.
More information: Ferdinand Greiss et al, A genetic circuit on a single DNA molecule as an autonomous dissipative nanodevice, Nature Communications (2024). DOI: 10.1038/s41467-024-45186-2
Journal information: Nature Communications
© 2024 Science X Network