This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Researchers uncover molecular mechanisms behind effects of MXene nanoparticles on muscle regeneration
Tissue engineering, which involves the use of grafts or scaffolds to aid cell regeneration, is emerging as a key medical practice for treating volumetric muscle loss (VML), a condition where a significant amount of muscle tissue is lost beyond the body's natural regenerative capacity. To improve surgical outcomes, traditional muscle grafts are giving way to artificial scaffold materials, with MXene nanoparticles (NPs) standing out as a promising option.
MXene NPs are 2D materials primarily composed of transition-metal carbides and nitride. They are highly electrically conductive, can accommodate a wide range of functional groups, and have stacked structures that promote cell interactions and muscle growth. While there have been practical demonstrations in the laboratory showcasing their ability to promote the reconstruction of skeletal muscles, the specific mechanism by which they do so remains unclear.
To address this gap, Associate Professor Yun Hak Kim from the Department of Anatomy and Department of Biomedical Informatics, alongside Professors Suck Won Hong, and Dong-Wook Han from the Department of Cogno-Mechatronics Engineering at Pusan National University developed nanofibrous matrices containing MXene NPs as scaffolds. They used DNA sequencing to reveal the genes and biological pathways activated by MXene NPs to aid in muscle regeneration.
These findings, published in Nano-Micro Letters, mark a significant advancement in the use of MXene scaffolds for treating muscle damage.
"This discovery posits a prospective avenue for the utilization of these materials to augment the efficacy of muscle tissue regeneration post-injury or damage," explains Professor Kim.
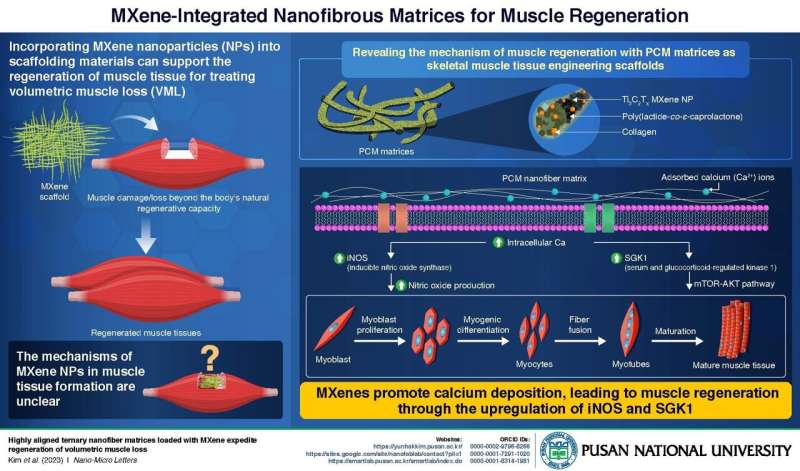
In the initial phase, the team created a nanofibrous PCM matrix containing poly(lactide-co-ε-caprolactone) (P), reinforced with collagen (C), and Ti3C2Tx MXene nanoparticles (M). To determine the specific effect of MXene NPs on muscle growth, they prepared three controls: pristine PLCL (P), PLCL with collagen (PC), and PLCL with MXene (PM). Upon testing all the scaffolds on mouse models with induced volumetric muscle loss, the researchers observed a significant increase in the overall number of muscle cells in PCM-treated mice compared to the other groups.
To understand how MXene nanoparticles (NPs) impact muscle regeneration and growth at the molecular level, the researchers introduced C2C12 myoblasts, which are precursors of muscle cells, onto PC and PCM matrices. The objective was to analyze the differences in gene expression levels between the two matrices. Within the PCM matrix, a heightened production of inducible nitric oxide synthase (iNOS) and serum/glucocorticoid-regulated kinase 1 (SGK1) was identified—two proteins closely associated with calcium signaling and muscle regeneration.
These results suggest that MXenes promote calcium ion (Ca2+) deposition around cells. This heightened levels of intracellular Ca2+ triggers the activation of genes that produce iNOS and SGK1 proteins. SGK1 influences the mTOR-AKT pathway, promoting cell proliferation, survival, and myogenesis—the conversion of myoblasts to muscle fibers. Simultaneously, iNOS increases the production of nitric oxide (NO), contributing to myoblast proliferation and muscle fiber fusion.
The combined effects lead to the development of mature muscle tissue. The aligned PCM nanofibrous matrices offer biophysical cues for intracellular biochemical signaling, guiding myogenic behaviors. This discovery contributes to our understanding of MXene's potential to regrow muscle and holds promise for refining scaffold designs to enhance this process further.
"Within 5 to 10 years, this research may yield groundbreaking treatments for muscle injuries. MXene NP-infused matrices could become a routine in medical practice for athletes, people with muscle-related ailments, and those recuperating from muscle-related traumas or surgeries," Prof. Kim states. "These NPs might enhance muscle regeneration methods, offering improved outcomes for reconstructive surgeries and conditions like muscular dystrophy, where muscle function is compromised."
The MXene NP-infused matrices hold potential for customization to meet diverse needs in treating muscle loss injuries. This customization may involve adjusting composition, structure, or properties to match specific patient requirements, like size, shape, or bioactivity enhancement. Tailoring these materials could offer personalized solutions for various muscle loss severities. Additionally, the observed enhanced muscle regeneration could aid in a more efficient recovery, potentially reducing post-treatment rehabilitation needs.
These matrices, with controllable mechanical properties, hold promise for enhancing in vivo muscle regeneration. Further research into MXene promises expanded clinical applications, potentially benefiting human well-being.
More information: Moon Sung Kang et al, Highly Aligned Ternary Nanofiber Matrices Loaded with MXene Expedite Regeneration of Volumetric Muscle Loss, Nano-Micro Letters (2024). DOI: 10.1007/s40820-023-01293-1
Provided by Pusan National University