This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Optogenetics study detects new acid sensor in plant cells
Using optogenetics, Würzburg researchers have detected a new acid sensor in plant cells that addresses a cell-internal calcium store. The study is published in the journal Science.
When plants are infected by pathogens, suffer from a lack of water or have to react to other external stimuli, the first thing they do is increase the proton and calcium concentration in the affected cells. The protons and calcium ions then act like messenger substances that trigger further reactions in the cell.
The interactions between protons and calcium ions in this process were previously largely unknown. The new study by a team led by biophysicist Professor Rainer Hedrich from Julius-Maximilians-Universität (JMU) Würzburg in Bavaria, Germany, has now shed new light on this subject.
Using a sophisticated optogenetic approach, the researchers have discovered a previously unknown endogenous acid sensor in plant cells. And they have discovered in the guard cells of leaves that there is a calcium store that plays an important role in processing proton signals in cellular responses.
Why such simple elements as protons and calcium ions act as signals
In the course of evolution, cells have designed their metabolism to utilize energy-rich phosphates. This results in a problem: At the predominantly neutral cellular pH value, the valuable phosphates can be bound by calcium ions (Ca2+) and converted into an insoluble and therefore no longer usable form (calcium dihydrogen phosphate).
To avoid this, cells keep their internal calcium level very low. In their environment, however, it is 10,000 times higher. Outside the cells, the concentration of protons (H+) and therefore the acidity is also much higher. Due to this concentration gradient, both types of ions have a strong urge to flow into the cells—making them ideal for use as messenger substances.
"The stimulus-dependent opening of calcium and proton channels in the cell membrane results in a temporary intracellular increase in both messenger ions," explains Hedrich. "The cells understand this as a signal, which they translate into a biological reaction using calcium- and proton-binding enzymes."
Light switch controls the flow of protons into the cell
How do plant cells react to the influx of protons and the associated acidification of their cell plasma? Until now, this could only be investigated with great experimental effort and even then only indirectly.
This is now much easier thanks to an appropriately equipped thale cress (Arabidopsis thaliana), which Hedrich's team has developed using optogenetic methods: A light-sensitive proton channel from a fungus, the channelrhodopsin KCR2, was optimized for use in plant cells. This means that protons can now be specifically sent into the cells in response to a light pulse. Furthermore, they expressed KCR2 together with the genetically encoded pH reporter pHuji. This makes it very easy to measure the current pH value in the cell upon KCR2 activation.
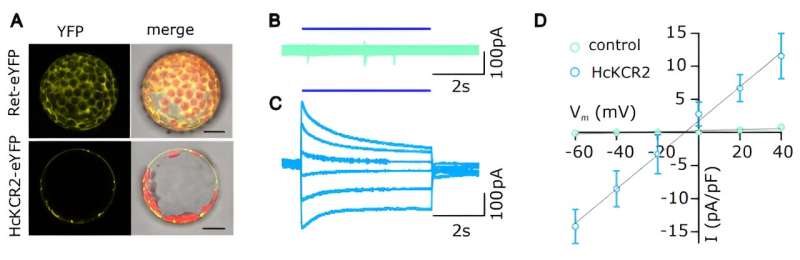
Shouguang Huang, the first author of the Science publication, next scrutinized the guard cells of the new Arabidopsis mutant. "When I stimulated them with blue light for a second, they depolarized, just as I had expected from a light-activated proton channel," says the researcher. During the subsequent experiments, the Würzburg ion channel specialists made a far-reaching discovery.
KCR2 activation acidifies the cell and causes calcium to rise
Their electrophysiological studies on guard cells showed that when the light stimulation began, the membrane potential immediately depolarized and the pH reporter pHuji signaled an acidification of the cell interior.
"However, we were astonished when the depolarization and acidification continued for a good minute after the end of the light pulse," says Hedrich. "This could only mean that the light activation of KCR2 and the acidification had activated the sphincter cell's own ion channels." These are the long-known guard cell anion channels SLAC1 and SLAH3, whose activation, however, also requires the presence of calcium.
Endoplasmic reticulum as a calcium store
"Taking all the facts together, it could be assumed that the proton currents carried by KCR2 and the associated acidification of the cell interior must also have generated a calcium signal," summarizes the JMU professor.
His team was able to prove that the rapid acidification of the guard cells is followed by a calcium signal that lasts for 150 to 200 seconds. And they discovered that this calcium does not come from outside the cell, but is released from an endogenous store, the endoplasmic reticulum. This is a network of membrane tubes and cisterns that run through the cytoplasm.
Future studies will now focus on analyzing the molecular nature of the H+-sensitive calcium channel of the endoplasmic reticulum and investigating its proton-activated on/off switch. Overall, these studies are important in order to better understand how plant cells react to external stimuli such as infections or drought.
More information: Shouguang Huang et al, Light-gated channelrhodopsin sparks proton-induced calcium release in guard cells, Science (2023). DOI: 10.1126/science.adj9696
Journal information: Science
Provided by Julius-Maximilians-Universität Würzburg