This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Malfunction in spermatogenesis: Researchers uncover contribution of cylicin proteins to male fertility
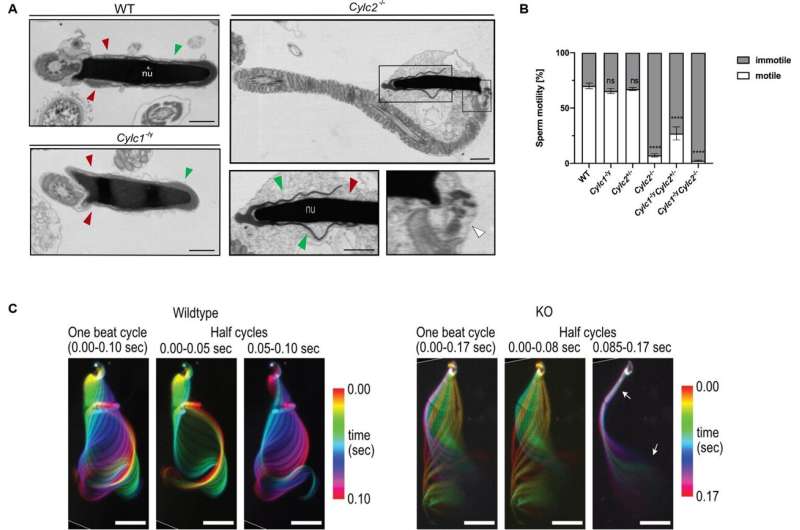
For successful fertilization, sperm should move forward rapidly and be shaped correctly. The unique structure of the sperm cells forms during spermiogenesis. Now, researchers from the University Hospital Bonn (UKB) and the Transdisciplinary Research Unit Life & Health at the University of Bonn have found that fertility problems in both mice and humans can be caused by loss of so-called cylicins. This causes defects in the head and tail structure of sperm. The results of the study have now been published in the journal eLife.
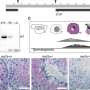
During spermiogenesis, round immature cells in the testes develop into mature sperm with a head and tail. At the front of the sperm head, which carries the genetic material, is the acrosome; a structure required to cross the protective layer around the egg during fertilization. The sperm tail allows the cell to actively reach the egg. The shape of the sperm is unique and the cytoskeleton contains special proteins found only in sperm cells.
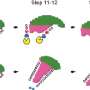
This cytoskeleton ensures the stable connection of the individual parts of the sperm. In the area of the sperm head there is the perinuclear theca (PT) which is a cytoskeletal structure surrounding the sperm head. In this PT Cylicin 1 and Cylicin 2 are located. The role of these proteins had not yet been investigated.
"Since cylicins are very similar in mice and humans, our study in mice also allows us to find out more about human sperm development," says corresponding author Prof. Hubert Schorle from the Institute of Pathology at UKB, who is also a member of the Transdisciplinary Research Area (TRA) Life & Health at the University of Bonn. "We hoped to gain insights into the complex network of factors that control sperm development and thus also male fertility."
Cylicins are a structural component of the sperm calyx
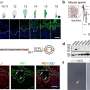
To find out where cylicins are needed in spermiogenesis, Prof. Schorle's research group created and studied mice that can no longer produce Cylicin 1 and/or Cylicin 2. "Male mice lacking Cylicin 1 achieved fewer pregnancies and smaller litters when mated with healthy females," says first author Dr. Simon Schneider of the Institute of Pathology and Bonn Technology Campus of the Faculty of Medicine.
First author Andjela Kovacevic, a Ph.D. student in Prof. Schorle's group, adds, "The absence of two or more copies of the cylicins rendered male mice infertile: mating resulted in no pregnancies."
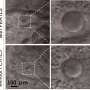
The reason for the observed infertility is that the sperm heads are smaller and the acrosomes are damaged. In addition, the tails are curled and wrapped around the sperm head, which massively restricts the mobility of the sperm. The Bonn researchers found that, an area of the PT called the calyx is missing in these sperm. "This suggests that the cylicins are essential for this sperm structure," Prof. Schorle says.
Cylicins also regulate male reproduction in humans
The Bonn researchers also wanted to find out whether fertility problems in humans are also partly due to changes in the cylicins. To this end, they teamed up with colleagues from the University of Münster. They were able to identify one patient in the patient cohort of the University Hospital Münster who has variants of Cylicin1 and Cylicin2 genes. His sperm has defects of the head and tail structure and the patient is unable to father a child.
"Based on these results, testing for gene variants of cylicins should be considered in cases of male infertility," says Prof. Schorle, who also has a hopeful message for those affected. "Since 'only' the scaffolding of the sperm is damaged, an attempt to transplant the sperm head, and thus the genetic material into an egg cell, might result in a successful fertilization."
More information: Simon Schneider et al, Cylicins are a structural component of the sperm calyx being indispensable for male fertility in mice and human, eLife (2023). DOI: 10.7554/eLife.86100
Journal information: eLife
Provided by University Hospital Bonn