This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
A promising approach in sustainable chemical production
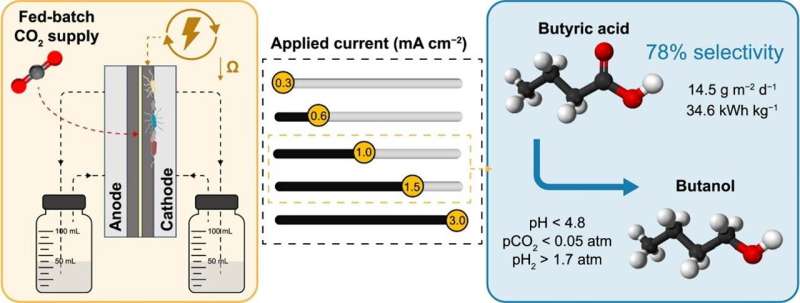
Carbon capture and utilization (CCU) technologies are crucial for addressing climate change while ensuring economic viability. MES has emerged as a promising approach for CO2 reduction to biofuels and platform chemicals. However, the industrial adoption of MES has been hindered by low-value products like acetate or methane and high electric power demand.
In a new study published in the journal Environmental Science and Ecotechnology, researchers from University of Girona conducted a study that focused on electrically efficient MES cells with low ohmic resistance (15.7 mΩ m2). Through a fed-batch mode, alternating high CO2 and hydrogen (H2) availability, they successfully promoted the production of acetic acid and ethanol.
Chain elongation resulted in the selective (78% on a carbon basis) production of butyric acid, a valuable chemical used in pharmaceuticals, farming, perfumes, and the chemical industry. At an applied current of 1.0 or 1.5 mA cm−2, the study achieved an impressive average production rate of 14.5 g m−2 d−1 of butyric acid. The key player in the chain elongation process was identified as Megasphaera sp.
Inoculating a second cell with the enriched community replicated the butyric acid production rate, but with an 82% reduction in the lag phase. Butyric acid was successfully upgraded to butanol, a valuable biofuel compatible with existing gasoline infrastructure and used as a precursor in pharmaceutical and chemical industries for acrylate and methacrylate production. Solventogenic butanol production was triggered at a pH below 4.8 by interrupting CO2 supply and maintaining specific pH and hydrogen partial pressure conditions.
The MES cell design proved highly efficient, with average cell voltages of 2.6–2.8 V and an electric energy requirement of 34.6 kWhel kg−1 of butyric acid produced. Despite some limitations due to O2 and H2 crossover through the membrane, the study identified optimal operating conditions for energy-efficient butyric acid production from CO2.
In conclusion, this study showcases the potential of bioelectrochemical conversion of CO2 to butyric acid and its subsequent upgrade to butanol in microbial electrolysis cells. The process holds promise for sustainable and economically viable production of valuable chemicals from CO2. Further research and development are crucial to optimize the process for large-scale applications, and with continued advancements, this technology can revolutionize chemical production while mitigating climate change impact.
More information: Meritxell Romans-Casas et al, Selective butyric acid production from CO2 and its upgrade to butanol in microbial electrosynthesis cells, Environmental Science and Ecotechnology (2023). DOI: 10.1016/j.ese.2023.100303
Provided by Chongqing Medical University