This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers reveal that the mechanism for forming colloidal gels differs from glass formation
The soft, solid-like properties of colloidal gels are essential in fields such as food and medical applications, but how these properties manifest themselves is a long-standing mystery. Until now, it was believed that the solid nature of gels emerges through glass formation.
In a study recently published in Nature Physics, researchers from the Institute of Industrial Science, The University of Tokyo have used a new microscopic technique—in situ confocal microscopy—to reveal the differences in gel and glass formation.
A colloidal liquid is a mixture consisting of small particles that are scattered throughout another liquid substance; milk is an example. Understanding how colloidal liquids can become a gel upon phase separation—through a so-called dynamically arrested state—is important for optimizing the design of foods, cosmetics, and biomedical materials. However, a single-particle-level explanation of the dynamically arrested state remains elusive.
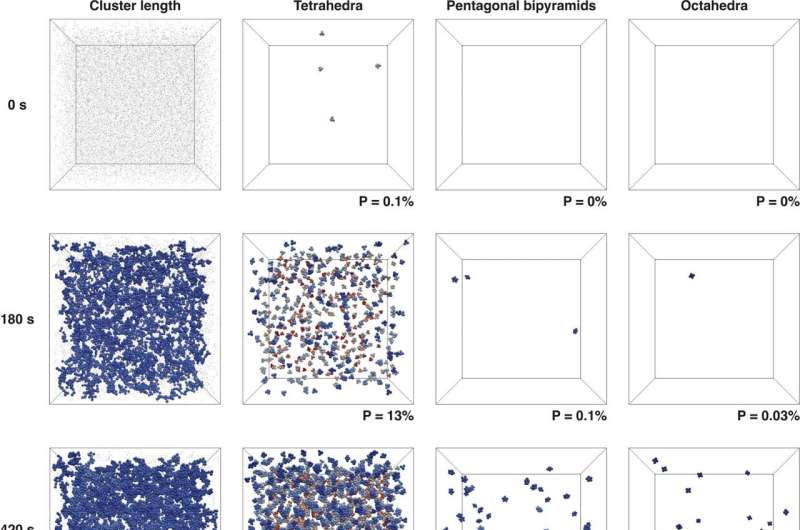
Efforts in this direction have recently focused on a principle known as local amorphous ordering, which pertains to the order of arrangement of the constituents of colloidal liquids. Nevertheless, there has not been an experimental means of visualizing such ordering, on a single-particle level, in the initial stage of colloidal gelation. Providing fundamental physical insights into the origin of amorphous ordering—and thus the dynamically arrested state—of colloidal gels is the problem that the researchers sought to address.
"A pentagonal bipyramid shape has a symmetry that is incompatible with crystallization, and might help prevent colloid particles from undergoing the gel-to-crystal transition," explains Hideyo Tsurusawa, lead author of the study. "We developed an in situ confocal microscopy method for testing this hypothesis in a real-time, real-space manner."
The researchers report results on dilute colloidal gels consisting of "sticky" spherical particles that exhibited short-range, directionless, attractive interactions. They revealed that different local particle arrangements uniquely modulated the properties of the gel. Specifically, tetrahedra arrest local particle motion, 3-tetrahedra hinder crystallization, and pentagonal bipyramid clusters impart solidity. The researchers propose that minimizing the local potential energy is central to forming a gel state, whereas minimizing the free energy (entropy) is central to forming a glass state.
"The unique feature characterizing formation of a dilute gel is a sequential hierarchical ordering from tetrahedra to pentagonal bipyramids and to their clusters," says Hajime Tanaka, senior author. "This is absent in glass formation."
This work proposed a novel mechanism based on sequential amorphous ordering for the dynamical arrest of colloidal gels, unlike the previous glass-based explanation, through directly accessing the gel formation process at a single-particle level. This work has practical implications in helping to optimize colloidal gel materials with desired mechanical properties; for example, in food and medical applications.
More information: Hideyo Tsurusawa et al, Hierarchical amorphous ordering in colloidal gelation, Nature Physics (2023). DOI: 10.1038/s41567-023-02063-x
Journal information: Nature Physics
Provided by University of Tokyo