This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Team successfully tests, validates new method for measuring the precise dimensions and comparability of biomolecules
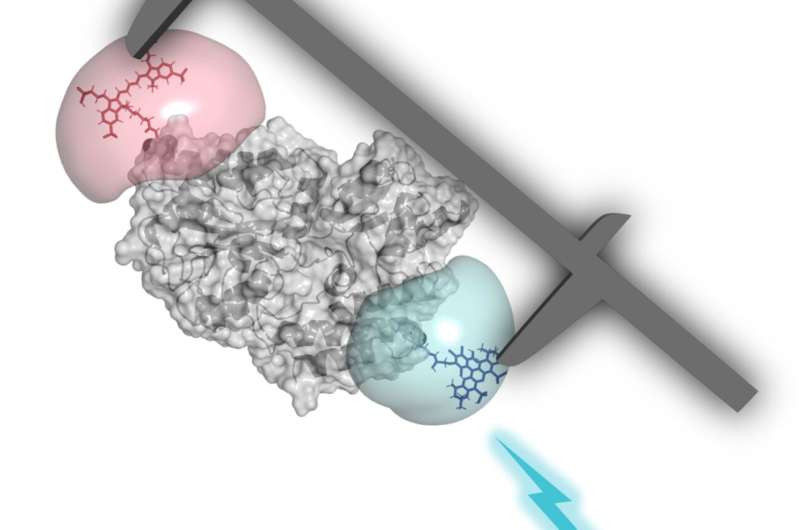
The precise measurement of biomolecules can play a critical role in improving our understanding of fundamental life processes. In a large-scale comparative study involving 19 laboratories around the globe, a team working with LMU scientists Professor Thorben Cordes and Professor Don C. Lamb, alongside Professor Claus Seidel of HHU in Düsseldorf and Dr. Anders Barth of Delft University of Technology, has now tested a method of measuring the precise dimensions and comparability of biomolecules.
Their findings are published in Nature Methods.
Proteins are the fundamental building blocks of life. Every animal, every plant and every microorganism is made up of proteins and only 'works' on the basis of countless complex processes that are controlled by the interplay of different proteins. It is therefore no wonder that science has a keen interest in a better understanding of these biochemical all-rounders.
The problem is that we cannot simply measure them with a ruler. Researchers thus have to resort to a whole toolbox full of different investigative methods in order to arrive at an accurate picture of what proteins look like, how they behave and how they work.
How do you measure moving protein structures?
The single-molecule FRET analysis is especially well suited to this purpose. It makes use of what is known as Förster resonance energy transfer (FRET), where energy from an excited chromophore is transferred without radiation to a second light-sensitive molecule. By artificially inserting color molecules (chromophores) in the biomolecules under investigation, it becomes possible to measure extremely small distances in the sub-nanometer range.

This approach already works quite well to measure distances between different molecules. The structure of DNA strands can likewise be examined fairly reliably. Compared to DNA, however, performing similar operations with proteins is considerably trickier. Proteins are more varied and, above all, more mobile, which makes them much more difficult to analyze.
Notwithstanding, the researchers conducting the study have now been able to establish the process for movable proteins too—successfully enough to achieve precise and reproducible results. For example, they were able to measure not only tiny distances within the protein complexes but also to observe structural differences as proteins changed their shape.
The laboratories taking part in the study were able to measure such structural changes to within one nanometer, and that on time scales of less than a millisecond. This astonishing precision shows that even dynamic protein systems can be reproducibly measured with FRET.
"Until now, many of our fellow structural biologists were skeptical about whether using FRET to analyze proteins could yield any reproducible findings at all, and about how to interpret results when proteins move," says Thorben Cordes. "We have now been able to dispel these doubts. But in doing so, we have also shown how tiny and how fast the movements of proteins can be for us to be able to observe and quantify them with FRET."
The researchers are convinced: Another versatile and reliable instrument has now been added to structural biologists' toolbox. Their hope is that the resultant data will also improve the accuracy of AI-based predictions and thereby further advance our understanding of dynamic processes in proteins.
More information: Ganesh Agam et al, Reliability and accuracy of single-molecule FRET studies for characterization of structural dynamics and distances in proteins, Nature Methods (2023). DOI: 10.1038/s41592-023-01807-0
Journal information: Nature Methods
Provided by Ludwig Maximilian University of Munich




















