This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Scientists explain how an infection can produce genetic diversity
As COVID has demonstrated, when pathogens are moving through the population, we adjust, limiting interactions, even isolating, and generally changing the way we associate with one other. Humans are not alone. New research from Harvard scientists provides some insight into how pathogens change animal social behaviors.
"Extreme environmental conditions have a very strong influence on all animals," said Yun Zhang, a professor in the Department of Organismic and Evolutionary Biology. But while this behavior has been seen in animals from simple fruit flies all the way up to primates, researchers have not understood what happens inside an individual animal's brain that leads to infection-induced changes in social behavior.
In their new paper, published in Nature, Zhang and colleagues studied the small roundworm C. elegans, which exists in nature with two sexes: hermaphrodites that produce both eggs and sperm, and males. Under normal conditions, the hermaphrodites are loners, preferring to self-reproduce over mating with males. However, Zhang's team found that the hermaphrodite worms infected by a pathogenic strain of the bacterium Pseudomonas aeruginosa became more interested in one another and increased their mating with males.
"In general, compared with self-reproduction, mating with males is more likely to produce novel genomes via recombination," added Zhang. "Therefore, pathogen-induced increase in mating strengthens the ability to produce genetic diversity for the adaptation of the host animals."
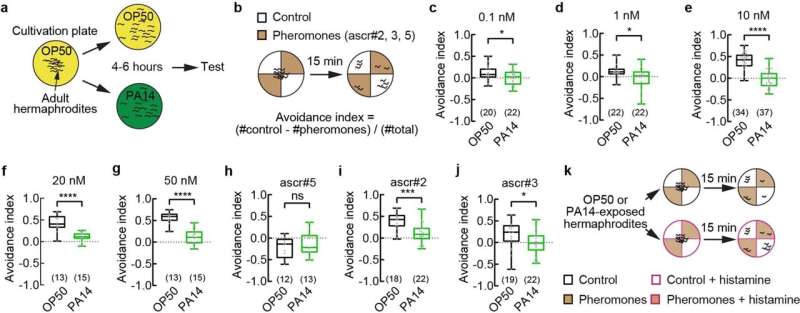
What drives this change in mating behavior? A mixture of pheromones—small volatile chemicals that are emitted by individual worms that other worms respond to—plays an important role.
"These pheromones are usually dispersing cues that make the hermaphrodites repel each other," said Tailhong Wu, a postdoctoral scholar in the Zhang lab and co-first author of the paper. But infected hermaphrodites become less repelled by the pheromones. Sometimes they are even attracted to them.
Specifically, the researchers found that one pair of chemical-sensing neurons in the worm began to respond to the pheromones after infection and that these neurons were needed for the worms to change their behavior.
Next, the researchers isolated messenger RNA from the pair of neurons, examining how they are different post-infection. They discovered that the pheromone receptor STR-44 was significantly upregulated in infected worms. The STR-44 receptor is a G-protein coupled receptor (GPCR), and its expression makes the pair of neurons respond to the pheromone mixture. The team tested many other pheromone receptors that were previously identified in worms, but none appeared to influence pathogen-induced social behavior change, suggesting STR-44's specific role in this process.
"Normally, expression of the STR-44 pheromone receptor is very low in the worms," said Minghai Ge, another postdoctoral scholar in the Zhang lab and co-first author of the paper. "But the exposure to the bacterial pathogen strongly induces the expression of this receptor." The presence of the larger amount of STR-44 pheromone receptor suppressed the repulsion of the hermaphrodite worms and increased their mating rate with males.
Looking beyond worms, Zhang pointed out that many different GPCRs for chemicals are encoded in the genomes of several animals. They are used to assess environmental cues, such as odors, tastes, and pheromones. Regulation of pheromone receptors may be a common strategy for animals to change their social behavior in the presence of a pathogen stress, she said.
"Animals have many GPCRs that can sense chemicals. It is possible that some of them are not normally used," Zhang said. "It's like they are usually saved in the bank, only to be used under stressful conditions, such as an infection."
The team think that the research provides a path for studying behavior change in response to pathogens and parasites in more complex animals. "This simple model animal gave us experimental powers to identify the neuronal and molecular basis for social behavioral plasticity," Zhang said.
Previous studies not from the Zhang lab have already identified the effects of pathogens on mating behavior of other invertebrate and vertebrate animals. "Perhaps other researchers can look at the pheromone responses important for mating behavior in these animals," she suggested, potentially explaining how infection affects the nervous system leading to behavioral changes, including those in social interactions.
More information: Taihong Wu et al, Pathogenic bacteria modulate pheromone response to promote mating, Nature (2023). DOI: 10.1038/s41586-022-05561-9
Journal information: Nature
Provided by Harvard University
This story is published courtesy of the Harvard Gazette, Harvard University's official newspaper. For additional university news, visit Harvard.edu.