This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Purifying water with just a few atoms

Due to their considerable efficiency, catalysts made of just a few atoms show great promise in the field of water treatment. In a new study, researchers looked into how to optimize the performance of these catalysts and make them viable for practical use.
The results of the study, led by Prof. Jaehong Kim, are published in the Proceedings of the National Academy of Sciences.
In the last few decades, nanoscale catalysts have drawn much attention in the field of water treatment. Materials at the nanoscale have numerous unique and beneficial properties to offer. More recently, researchers have been exploring the possibilities of single-atom catalysts. Even smaller than nanomaterials, these catalysts can offer even greater efficiency.
"We didn't have this capability before, but now we are basically loading single-atom metals, atom by atom, onto the substrate," said Kim, the Henry P. Becton Sr. Professor of Chemical & Environmental Engineering. "And that's great, because you can utilize all of the atom."
Efficiency is critical because the materials that are commonly used for catalysts can be very expensive. For instance, palladium (currently going for about $2,000 per ounce) is a metal that's often used for catalysts. A quick comparison shows why single-atom catalysts have generated so much interest. In nanoscale form, 50 nanometers of palladium cost about $37 to cover an area of about 250 square meters. Just over 2% of its atoms are exposed at the surface. In single-atom form, in contrast, palladium costs only 17 cents to cover an area greater than 50 football fields. And you can't beat it for efficiency, with 100% atom exposure at the surface.
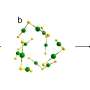
One limitation of the single-atom catalysts is that certain conditions can diminish their catalytic performance. As a solution to that, researchers have now begun creating catalysts comprising a small cluster of atoms, known as ensembles. Instead of the thousands of atoms that make up a nanomaterial, these clusters contain just three or four atoms. "But they exhibit properties more like a single atom because they're such a small cluster, and the atoms are all exposed at the surface," Kim said.
Because this material design is still relatively new, researchers are still figuring out the best ways to control the properties of these ensemble structures and optimize their performance. For instance, fully isolated single-atom catalysts can be enhanced by the addition of certain elements around the metals. Kim and his research team investigated whether atom ensembles could be similarly manipulated. Their paper is the first to explore the possibilities of doing so.
Kim created a system with a catalyst using an ensemble of palladium atoms, designed to reduce the carcinogen bromate in water. They introduced the non-metal elements sulfur, nitrogen, and boron to the surrounds of atom ensembles. The overall results suggested an improvement in the system's catalytic performance. It's a promising sign, Kim said, especially since water treatment needs to be as cost-effective as possible.
"Ultimately, we are hoping to have a highly efficient device that has this catalyst to destroy pollutants in water, because it is going to be so much cheaper and efficient than other material designs," he said.
More information: Dahong Huang et al, Enhancing the activity of Pd ensembles on graphene by manipulating coordination environment, Proceedings of the National Academy of Sciences (2023). DOI: 10.1073/pnas.2216879120
Journal information: Proceedings of the National Academy of Sciences
Provided by Yale University