This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Deep learning enhanced NIR-II volumetric imaging of whole mice vasculature
In vivo small animal fluorescence imaging is widely used in oncology, drugs, antibodies, nanomaterials, inflammation, immune diseases, gene therapy, apoptosis, and other research fields because of its advantages such as in vivo real-time imaging observation of labeled animal tissues or exogenous drugs.
This system allows for the long-term follow-up of the growth, metastasis, and drug treatment process of tumors in living animals, the development process of infectious diseases, inflammation and bone damage repair, the expression process of specific genes, and the metabolism process of nanomedicine in the organism. The system is also an imaging platform for the design and optimization of various fluorescent materials and biological probes.
Near-infrared II (NIR-II: 1000-1700 nm) is a longer wavelength photon than visible light and NIR-I light. It is less scattered in biological tissues and has lower background interference from biological tissue autofluorescence.
With the development of NIR-II photoluminescent probes and supporting cameras and other equipment, the application of NIR-II fluorescence in vivo functional imaging has become a research hotspot. However, current NIR-II in vivo imaging systems are still based on planar imaging obtained with industrial lenses, which can obtain millimeter-depth fluorescence imaging but cannot reflect three-dimensional depth information.
In addition, mice are far away from the lens, and the imaging resolution is low. The effect of tissue scattering still exists. Therefore, the development of deep, high-resolution, spatial three-dimensional, high-contrast in vivo imaging systems has become an important development direction for in vivo fluorescence imaging of small animals.
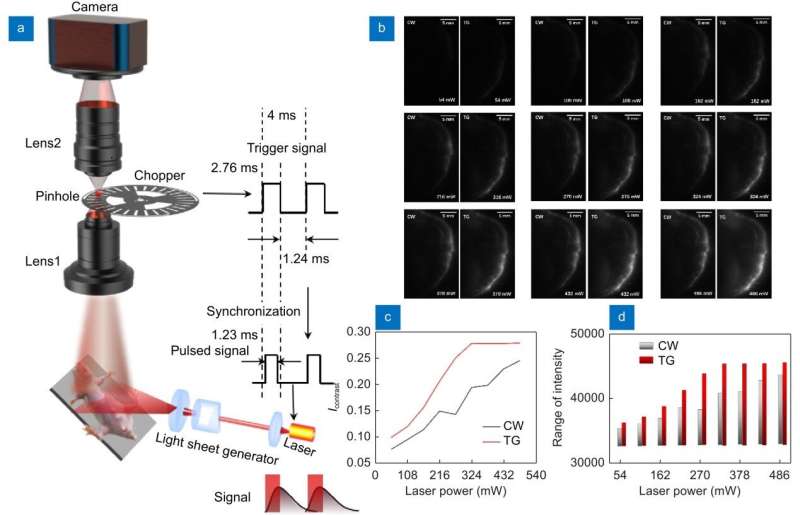
Light-sheet fluorescence microscopy imaging is a three-dimensional imaging technique applied to cells, organoids, and small embryos that allows for rapid three-dimensional imaging of biological samples. In order to achieve deep 3D in vivo imaging with NIR-II fluorescence, improve imaging resolution, and reduce the influence of fluorescence scattering and tissue autofluorescence, the team led by academician Dayong Jin from Southern University of Science and Technology prepared a NIR-II probe with rare earth nanoparticles to detect fluorescence with a peak of about 1530 nm.
They first introduced light-sheet imaging into the field of adult whole mouse in vivo imaging. The team used time-gating technology to reduce thermal effects and average laser power. They also enlarged the numerical aperture of imaging by coupling imaging with dual industrial lenses and developed a vascular enhancement algorithm based on deep learning to improve imaging contrast. These advancements enabled high-resolution whole-mouse vascular network imaging in the NIR-II region with deep 3D in vivo imaging.
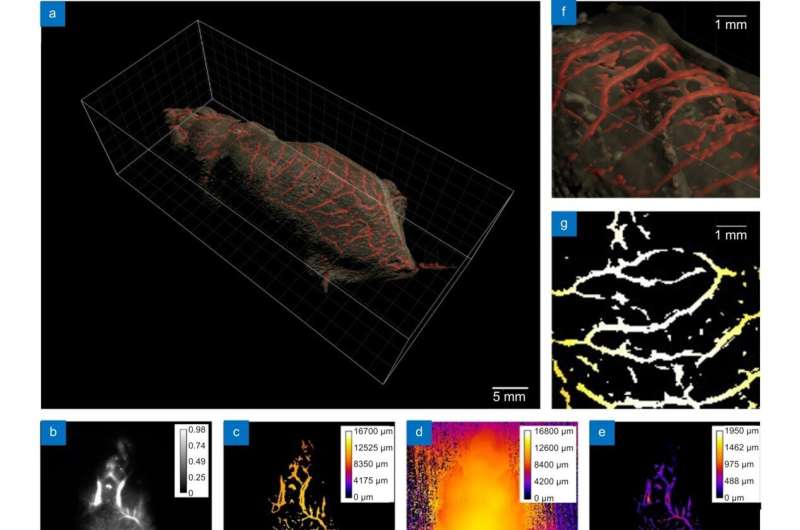
This study achieved clear resolution of blood vessels in deep tissue with NIR-II body imaging through the synergistic use of time-gated imaging, light sheet scanning, and deep learning algorithms. These techniques effectively reduced the effects of laser scattering and out-of-focus background noise.
The 3D in vivo imaging system successfully reconstructed the full length of 527.7 mm of blood vessels in the macroscopic whole mouse subcutaneous 2 mm depth range. The smallest resolvable vessel diameter was up to 100 µm, and the depth resolution was 100 µm. This technique fills the gap between conventional microscopic imaging and macroscopic imaging, providing the possibility of 3D whole-body pathology model research.
The system rapidly acquires massive amounts of information and can analyze microscopic details of macroscopic regions of interest, such as the transport processes of cancer cells in blood vessels, in the context of the complete human body. The new generation of NIR-II 3D in vivo deep HD imaging offers significant advantages over current tissue transparency techniques used to avoid scattering effects.
The work is published in the journal Opto-Electronic Advances.
More information: Sitong Wu et al, Deep learning enhanced NIR-II volumetric imaging of whole mice vasculature, Opto-Electronic Advances (2022). DOI: 10.29026/oea.2023.220105
Provided by Compuscript Ltd