This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers discover mechanism that contributes to Salmonella replication, informs other bacterial infections
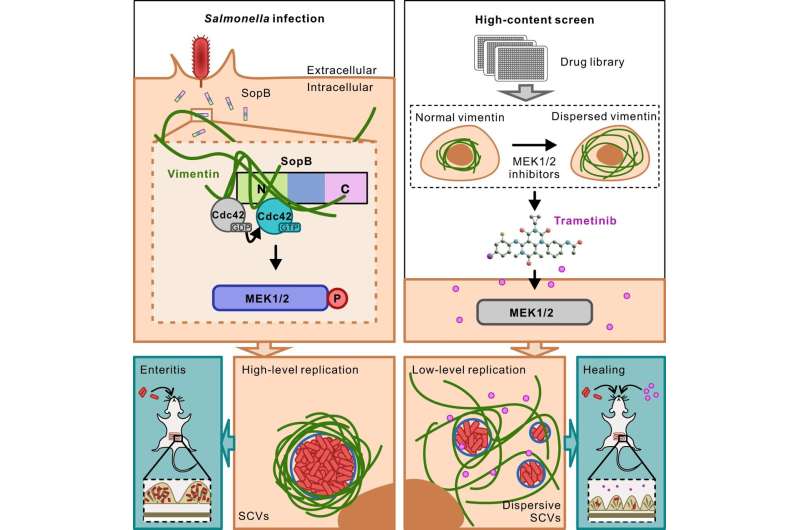
In a study published in Nature Communications, Prof. Jiu Yaming's group at Institut Pasteur of Shanghai of the Chinese Academy of Sciences reported a previously unrevealed function of intermediate filaments that protect bacterial replication via constructing a cage embracing the replication machinery.
Salmonella is a major food-borne pathogen that causes millions of gastrointestinal and systemic diseases globally each year. Upon invasion into host cells, the majority of Salmonella replicates in a membrane-bound compartment known as the Salmonella-containing vacuole (SCV).
Emerging evidence suggests that cytoskeletal actin filaments reassembling and microtubule motor-based movement are crucial host cytoskeletal machinery for the oriented migration and positioning of SCV to the perinuclear region, for efficient replication. However, the role of intermediate filaments (IFs), which are crucial for mechanical strength and resilience of the cell, in bacterial vacuole preservation remains unclear.
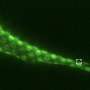
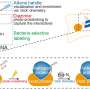
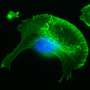
Here, Prof. Jiu's group discovered that Salmonella effector SopB reorganizes the vimentin network to form cage-like structures that surround SCVs. Genetic removal of vimentin markedly disrupted SCV organization, significantly reduced bacterial replication and cell death. Mechanistically, SopB used its N-terminal Cdc42-binding domain to interact with and activate Cdc42 GTPase, which in turn recruited vimentin around SCVs. A high-content imaging-based screening identified that MEK1/2 inhibition led to vimentin dispersion.
The researchers therefore elucidated the signaling axis SopB-Cdc42-MEK1/2 as mobilizing host vimentin to maintain concrete SCVs and identified a decisive mechanism contributing to Salmonella replication. Importantly, a clinically-approved drug identified in the screen, displayed significant anti-infection efficacy against Salmonella both in vitro and in vivo.
The identified interplay between vimentin and SopB provides a new approach for future mechanistic studies focusing on other Gram-negative intracellular bacterial infections. In addition, the researchers repurposed the drug identified as a possible anti-bacterial treatment for salmonellosis, which may help to reduce the abuse of antibiotics, and expand the anti-infectious spectrum of the drug.
More information: Shuangshuang Zhao et al, Salmonella effector SopB reorganizes cytoskeletal vimentin to maintain replication vacuoles for efficient infection, Nature Communications (2023). DOI: 10.1038/s41467-023-36123-w
Journal information: Nature Communications
Provided by Chinese Academy of Sciences