This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Using cancer cells as logic gates to determine what makes them move

Cancer cells migrate through the body for multiple reasons; some are simply following the flow of a fluid, while others are actively following specific chemical trails. So how do you determine which cells are moving and why? Purdue University researchers have reverse-engineered a cellular signal processing system and used it like a logic gate—a simple computer—to better understand what causes specific cells to migrate.
For many years, professor of mechanical engineering Bumsoo Han and his research group have been studying cancer cells. He builds microfluidic structures to simulate their biological environment; he has even used these structures to build a "time machine" to reverse the growth of pancreatic cancer cells.
"In our experiments, we've been observing and studying how these cancer cells migrate, because it's an important aspect of cancer metastasis," said Hye-ran Moon, postdoctoral researcher on Han's team. "But this is different. We are trying to address the fundamental mechanisms behind these behaviors. And it's very challenging because cells are very complex systems of molecules, and they are exposed to multiple cues that cause them to move."
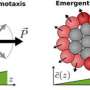
One of those cues involves chemical trails, which many cells are inherently drawn to (much like ants following a scent trail). Another is fluid flow; if fluids are flowing around cells in a certain direction, many cells will just go along for the ride. So if a cell is moving, how can you tell if it's motivated by chemicals, fluid motions, or both?
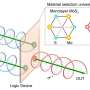
The team adopted a ternary logic gate model to analyze these cues, and predict how cells would move under different environments. Their research has been published in the journal Lab on a Chip.
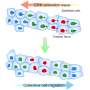
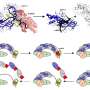
Their experiments took place in a microfluidic platform with a center chamber for the cells, and two side platforms. Using this device, they could replicate fluidic flows in one direction, in the opposite direction, or no flow at all. They could also introduce a chemical known to cause the cells to migrate. Again, they had the option of chemotaxis in one direction, the opposite direction, or none at all. Would these two cues multiply, or cancel each other out?
"With two cues and three choices each, we had enough observable data to build a ternary logic gate model," said Moon.
Logic gates are a construct from computing, where transistors take a 1 or 0 input and return a 1 or 0 output. Binary logic gates take a combination of two 1s and 0s, and output different results based on what kind of gate it is. Ternary logic gates do the same thing, except with three possible inputs and outputs: 1, 0, and -1.
Moon assigned values to which direction the cells moved under the two different stimuli. "If the cells moved in the direction of the flow, that's 1," said Moon. "If they have no directionality, that's 0. If they move in the opposite direction to the flow, that's -1."
When cells encountered either chemicals or fluid flow individually, they moved in the positive direction (the "1"). When both were present in the same direction, the effect was additive (still "1"). However, when the two flowed in opposite directions, the cells moved in the direction of the chemicals (the "-1"), rather than the fluid flow.
Based on these observations, Moon extrapolated a 3x3 grid to simplify the results. The cues of these cancer cells could now be diagrammed much like an electrical engineer would diagram a circuit.
Of course, the real world is never that simple. "In actuality, the chemical stimulus is a gradient, not an on-off switch," said Moon. "The cells will only move once a certain threshold of flow has been introduced; and if you introduce too much, the cell short-circuits and doesn't move at all. The accuracy with which we can predict that movement is a non-linear relationship."
Moon also stressed that this particular experiment is very simple: two stimuli, in strictly opposite directions, in a single dimension. The next step would be to build a similar experiment, but in a 2-dimensional plane; and then another in a 3-dimensional volume. And that's just for starters; once you add multiple stimuli, and factor in time as the 4th dimension, the calculations become incredibly complex. "Now you understand why biologists need to use supercomputers," said Moon.
This study was in collaboration with the Purdue Institute for Cancer Research; the Weldon School of Biomedical Engineering; the Purdue Department of Physics and Astronomy; and Andrew Mugler and Soutick Saha of the University of Pittsburgh Department of Physics and Astronomy.
"This is a perfect example of how microfluidic devices can be used in cancer research," said Moon. "Doing this experiment in a biological environment would be extremely difficult. But with these devices, we can go right down to individual cells and study their behavior in a controlled environment."
"This model can apply to far more than just physical cancer cells," Moon continued. "Any cells can be impacted by different cues, and this provides a framework for researchers to study those influences and determine why they happen. Genetic engineers also have embraced the logic gate model, treating genes as processors that give different results when you give them certain instructions. There are many branches we can go with this concept."
More information: Hye-ran Moon et al, Cells function as a ternary logic gate to decide migration direction under integrated chemical and fluidic cues, Lab on a Chip (2022). DOI: 10.1039/D2LC00807F
Journal information: Lab on a Chip
Provided by Purdue University