G-protein coupled receptor findings could accelerate research and development of new cancer treatments
Researchers from City of Hope, one of the largest cancer research and treatment organizations in the United States, have identified how a protein receptor targeted by about 33% of all federally approved medication works. The discovery could facilitate pharmaceutical research because how and why this protein chooses to link to other proteins is critical to how cells will respond to medicines.
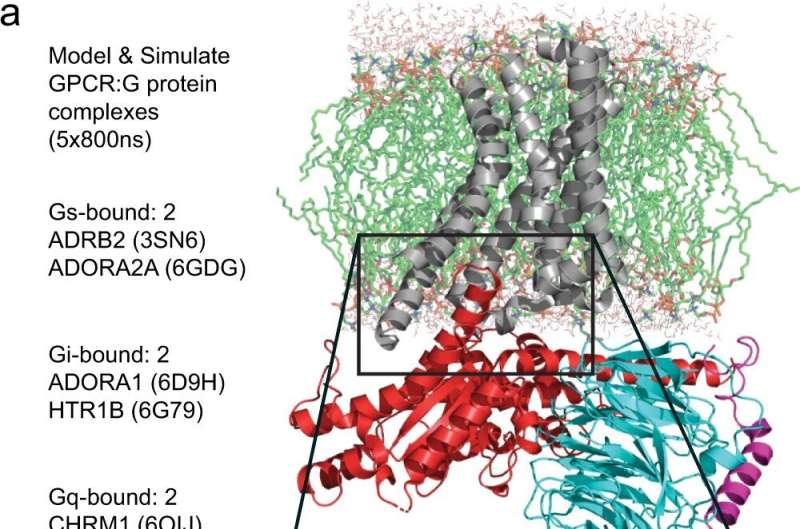
The recently published Nature Communications study, "Dynamic spatiotemporal determinants modulate GPCR:G protein coupling selectivity and promiscuity," uncovered a cell signaling mechanism of the largest superfamily of drug target proteins, called G-protein coupled receptors. Located in the cell membrane, GPCRs bind to substances outside the cell, which in turn initiates coupling of GPCRs to G proteins, leading to biological changes in the cell. Mutations in GPCRs and G proteins have been implicated in cancer and diabetes.
"Using large-scale data analysis techniques, we have identified the rules of engagement of GPCRs with G proteins—or the 'QR code' for their coupling," said Nagarajan Vaidehi, Ph.D., senior author of the study and chair of the Department of Computational and Quantitative Medicine at City of Hope. "Understanding the coupling mechanism of GPCRs to G proteins will aid in the design of drugs with lower side effects that target cancer-associated mutations."
Researchers can use the QR code to design small molecule drugs that specifically affect the coupling of a target GPCR to a specific G protein. Such a drug will affect only the cell signaling pathways that are cancer-associated and leave the other pathways unaffected. This process would lead to cleaner and more targeted cancer therapies.
"We are using the QR code to identify more target-specific drugs," Vaidehi said. "These findings are generalizable to any protein-to-protein coupling, which are emerging targets in personalized medicine," Vaidehi said.
More information: Manbir Sandhu et al, Dynamic spatiotemporal determinants modulate GPCR:G protein coupling selectivity and promiscuity, Nature Communications (2022). DOI: 10.1038/s41467-022-34055-5
Journal information: Nature Communications
Provided by City of Hope National Medical Center