Shortening the juvenile period for citrus crops to improve food stability
Citrus represents important commercial fruit crops worldwide. The juvenile period of citrus ranges from 6 to 20 years, seriously hindering conventional breeding and heredity improvement. Furthermore, the selection of ideal commercial and cultural traits through traditional breeding methods is time consuming. Shortening of the juvenile period and early flowering have therefore always been important breeding goals for citrus. Previous studies have confirmed that Flowering Locus T (FT) protein acts as a florigen, a mobile flower-inducing signal that is synthesized in leaves and then transported to meristems. Several studies have shown that citrus FT can shorten the juvenile period and participate in seasonal flowering. However, it remains unclear whether these FT proteins or mRNAs are mobile in citrus and can shorten the juvenile period.
In recent work published in the journal Horticulture Research, Huazhong Agricultural University researchers reported the movement of FT protein as a systemic signal in trifoliate orange (Citrus trifoliata) and its low accumulation in grafted juvenile scions.
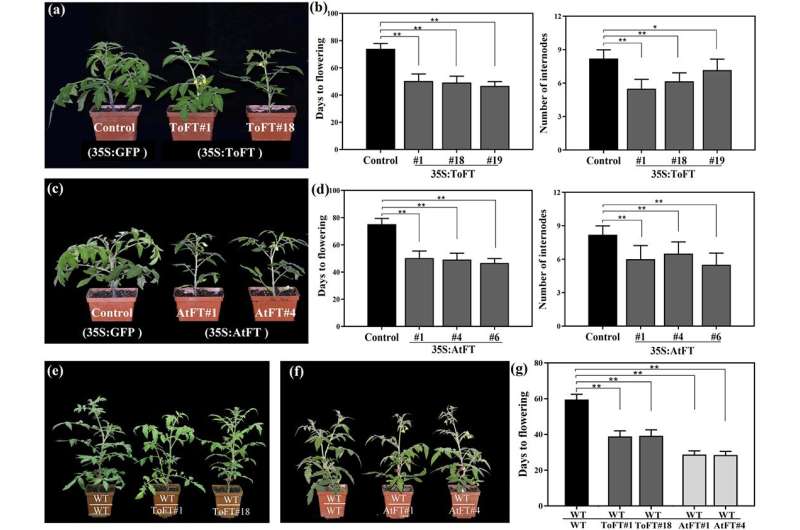
They first overexpressed FT genes from Arabidopsis (AtFT) and trifoliate orange (ToFT) in tomato and trifoliate orange to analyze the biological functions of AtFT and ToFT. The results showed that these two FT genes induced flowering in two types of transgenic plants (transgenic tomato and transgenic trifoliate orange). The authors then used the early-flowering transgenic plants as rootstocks/scions and wild-type plants as the corresponding scions/rootstocks in grafting experiments to further confirm that FT proteins can be transported over long distances and that FT transport is conserved between annual and perennial plants.
Wild-type tomato scions were grafted onto 35S:AtFT and 35S:ToFT transgenic tomato rootstocks to promote early flowering of the scions. However, when 35S:ToFT and 35S:AtFT transgenic trifoliate orange were used as rootstocks, the transport of FT protein did not significantly change the flowering time of non-transgenic scions during the two years of observation. Further analysis showed that when wild-type juvenile scions were grafted onto 35S:AtFT or 35S:ToFT transgenic rootstocks, the grafted scions showed significantly reduced endogenous ToFT protein, compared with juvenile trifoliate orange; therefore, total ToFT protein did not accumulate in significant amounts in the grafted scions. These results implied that the exogenous AtFT or ToFT protein may cause a decrease in the endogenous ToFT protein, resulting in the failure of the total ToFT protein to reach the threshold required for trifoliate orange flowering.
The authors also found that ToFT and AtFT could interact with ToFTIP1 in yeast two-hybrid (Y2H), bimolecular fluorescence complementation (BiFC), and pull-down assays. Grafting analysis has shown that AtFT and ToFT can be transported in trifoliate orange and tomato, respectively, and the authors also confirmed that AtFT and ToFT could interact with tomato SlFTIP1 in Y2H, BiFC, and pull-down assays. Overexpression of ToFTIP1 in tomato resulted in early flowering of the transgenic plants.
Grafting is an important tool for improving the production of horticultural crops. For citrus cultivation, the selection of appropriate rootstocks can improve nutrient absorption, plant vitality, stress resistance, and yield. Trifoliate orange is commonly used as a rootstock in citrus cultivation. Therefore, early-flowering transgenic trifoliate orange rootstocks generated in future work could provide a helpful and safe tool to dramatically accelerate citrus genetic studies and breeding.
More information: Yan-Mei Wu et al, Mobility of FLOWERING LOCUS T protein as a systemic signal in trifoliate orange and its low accumulation in grafted juvenile scions, Horticulture Research (2022). DOI: 10.1093/hr/uhac056
Provided by Nanjing Agricultural University The Academy of Science