'Youngest' antibiotic kills bacteria via a new two-step mechanism
Scientists at Utrecht University have discovered a new mechanism antibiotics use to kill bacteria. The antibiotic teixobactin uses a dual molecular strategy: it blocks the bacterial cell wall synthesis and destructs the cell membrane, the researchers write in the scientific journal Nature. The new insights will enable the design of powerful antibiotics against which bacteria do not readily develop resistance.
Antibiotics are used to treat bacterial infections and are among the most widely used drugs worldwide. They are vital to combat many infections in the respiratory and intestinal tracts, and various skin conditions. However, bacteria have become increasingly resistant to antibiotics, as most antibiotic classes currently used in clinics have been in use for about half a century. In 2015, scientists from Boston (U.S.) succeeded in isolating teixobactin, the first novel antibiotic discovered in about 40 years.
Isolated from 'unculturable' bacteria
Teixobactin came from so-called "unculturable" bacteria that do not grow under conventional laboratory conditions and shows extraordinary efficacy against bacterial pathogens without detectable resistance. However, how teixobactin killed bacteria was not understood. Most antibiotics bind to the ribosome, and by doing so, they inhibit the protein production of the bacteria. But there are also antibiotics that hinder bacteria's production of peptidoglycan, a sort of protective envelope that surrounds the bacteria. Teixobactin also does this, it soon appeared, but not in the usual way.
Long fibrils
Now, seven years after its discovery, a large international team led by Associated Professor Markus Weingarth from Utrecht University has discovered how teixobactin kills bacteria at a molecular level. The team used several advanced structural biology techniques and collaborated with the discoverers of teixobactin Prof. Kim Lewis of Northeastern University in Boston and the company Novobiotic.
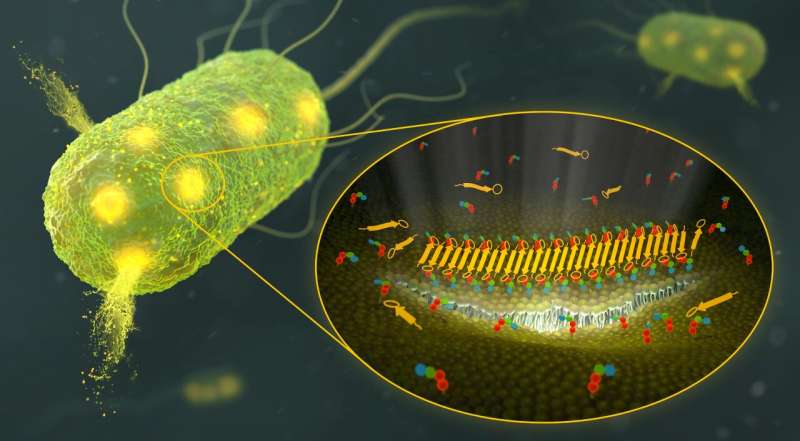
"Bacteria need a special lipid called lipid II to build their protective envelope around them. We show at atomic level that teixobactin targets and sequesters lipid II. Afterwards, teixobactin and lipid II together form long fibrils on bacterial cell membranes," says Weingarth. "Gradually, this creates a kind of valley in the landscape of the cell membrane, which then breaks down and damages the membrane." The Utrecht researchers turned out to have found a biomolecular mechanism that was unknown to science until now. "It is a unique killing strategy."
Better drugs against certain respiratory diseases
Ph.D. candidate Rhythm Shukla, who is the first author of the study, and Weingarth used the technique solid state nuclear magnetic resonance (ssNMR) to solve the complex formed by teixobactin and lipid II at atomic level in lipid membranes. Subsequently, the team was able to visualize the fibrils in membranes and directly in bacteria using different microscopy techniques.
Weingarth: "This is a first time to use this powerful approach to elucidate the action of membrane-active antibiotics. Antibiotics that act on the cell membrane are very difficult to understand. Now that we know how teixobactin works, it will be possible to rationally design better drugs against pneumonia, tuberculosis, or infections with MRSA bacteria. For all of these respiratory diseases, resistance to current antibiotics is a huge problem."
More information: Markus Weingarth, Teixobactin kills bacteria by a two-pronged attack on the cell envelope, Nature (2022). DOI: 10.1038/s41586-022-05019-y. www.nature.com/articles/s41586-022-05019-y
Journal information: Nature
Provided by Utrecht University Faculty of Science