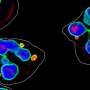
Study identifies way to specifically target and block disease-associated white blood cells
Neutrophils are a type of white blood cell that helps fight illness and disease by traveling to the body's infected site to seek and destroy harmful pathogens.
But left unrestricted, neutrophils can also prolong inflammation and contribute to the development of conditions like vascular thrombosis, cancer and diabetic retinopathy.
To block the defensive cell's harmful effects, a research team led by Case Western Reserve University (CWRU) has designed a nanoparticle platform that can exclusively target disease-associated activated neutrophils—while leaving inactive circulating neutrophils untouched.
"It ensures that the disease-associated neutrophils are suppressed," CWRU professor Evi Stavrou explained, "but [their] ability to fight infections remains intact."
The findings offer the potential to transform therapies for prevalent diseases, including diabetic complications, cancer and autoimmune disorders.
Stavrou, the Oscar D. Ratnoff Designated Professor in Medicine and Hematology at the School of Medicine, is lead corresponding author on the study, which recently appeared in Nature Nanotechnology.
The results emerged from a three-year collaboration among Stavrou's lab, colleagues at Case Western Reserve's biomedical engineering and pharmacology departments and with research partners around the world.
"This collaboration between Dr. Stavrou's lab and our lab brings our complementary and interdisciplinary expertise together to create a unique nanomedicine platform that enables specific targeting of activated neutrophils," said Anirban Sen Gupta, professor of biomedical engineering and the Leonard Case Jr. Professor of Engineering at the Case School of Engineering. "Aberrant neutrophil activity is emerging as a major mechanism in many diseases, and this platform can allow targeted treatment of such diseases without compromising neutrophil's immune defense capabilities."
Their work showed that targeting a neutrophil-suppressing drug specifically to the disease site by packaging it in the nanoplatform increased the drug's effectiveness. Doing so also decreased the toxic effects compared to when the drug is directly administered intravenously.
The study
The study represents the first demonstration of active targeting of what are called neutrophil "sub-populations." Their platform is versatile enough to be tailored to specific neutrophil populations—alone or in cell complexes, Stavrou said.
To target activated neutrophils specifically, Stavrou and Sen Gupta first had to identify a surface marker uniquely expressed by activated neutrophils but not by resting cells. They focused on neutrophil elastase (NE)—which is secreted by neutrophils during inflammation—because it's only produced by neutrophils and only travels to the cell's surface when activated.
To use NE as "bait" for nanoparticle (NP) binding, Stavrou and Sen Gupta designed a peptide derived from alpha-1 antitrypsin (a natural inhibitor of NE) and demonstrated its specific binding ability toward NE. Decorating the nanoparticle surface with this peptide enabled its specific binding to activated neutrophils.
Next, pharmacologic inhibitors that interfere with neutrophil functions were chosen. Combining these two components on a lipid NP platform generated active neutrophil-targeted therapeutic nanoparticles.
The assembled nanoparticles were used in in vitro and in vivo tests to define their loading ability, biodistribution, specificity toward NE and circulation lifetime in mouse models.
Several variations of NPs were created that can specifically interact with activated neutrophils only, or with activated neutrophils that are in complex with other cells such as activated platelets—a hallmark of inflammatory thrombosis in many diseases.
Finally, the feasibility of "targeted therapeutic capability" with this NP platform was demonstrated using drug payloads in mouse models of venous thrombosis.
The next phase of studies will focus on investigating new neutrophil-suppressing drug molecules developed in Stavrou laboratory as payload in the nanoparticles and evaluating these formulations in various neutrophil-driven disease models.
More information: Michelle A. Cruz et al, Nanomedicine platform for targeting activated neutrophils and neutrophil–platelet complexes using an α1-antitrypsin-derived peptide motif, Nature Nanotechnology (2022). DOI: 10.1038/s41565-022-01161-w
Journal information: Nature Nanotechnology
Provided by Case Western Reserve University