How gas nanobubbles accelerate solid-liquid-gas reactions
Solid-liquid-gas reactions are common in various natural phenomenon and industrial applications, such as hydrogen-oxygen fuel cell reactions, heterogeneous catalysis and metal corrosion in ambient environments. However, the gas transport in liquid and following reactions at the triple-phase interfaces are not well understood.
A joint research team led by Prof. Chen Jige from the Shanghai Advanced Research Institute (SARI) of the Chinese Academy of Sciences reported a real-time observation of the accelerated solid-liquid-gas etching progress of gold nanorods by introducing gas nanobubbles. They found that the underlying microscopic mechanism was dependent on liquid layer thickness.
The results were published in Nature Materials.
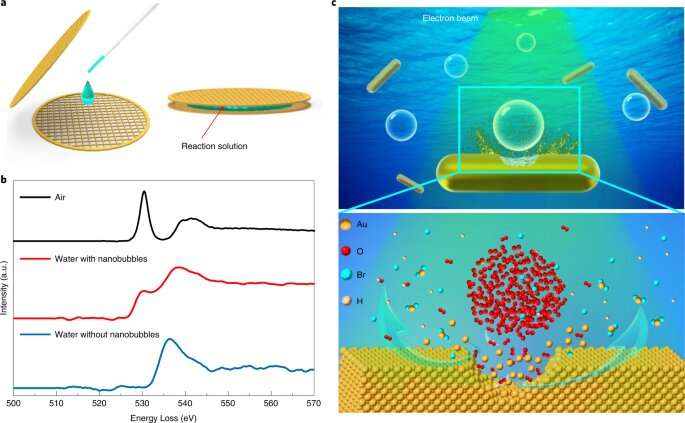
Liquid-cell transmission electron microscopy (TEM) enables the real-time observation of the accelerated etching of gold nanorods with oxygen nanobubbles in aqueous hydrobromic acid.
The researchers found that when an oxygen nanobubble was close to a nanorod below the critical distance (~1 nm), the local etching rate was significantly enhanced by over one order of magnitude.
Molecular dynamics simulation results revealed that the strong attractive van der Waals interaction between the gold nanorod and oxygen molecules governed oxygen transport through the thin liquid layer and thus led to enhanced etching rate.
This study sheds light on the rational design of solid-liquid-gas reactions for enhanced activities and provides a promising approach to modify the solid-liquid-gas reaction rate.
More information: Wen Wang et al, Solid–liquid–gas reaction accelerated by gas molecule tunnelling-like effect, Nature Materials (2022). DOI: 10.1038/s41563-022-01261-x
Journal information: Nature Materials
Provided by Chinese Academy of Sciences