New catalysts for better fuel cells

Researchers at Daegu Gyeongbuk Institute of Science & Technology (DGIST) have developed nano-catalysts that can reduce the overall cost of clean energy fuel cells, according to a study published in the journal of Applied Catalysis B: Environmental.
Polymer electrolyte membrane fuel cells (PEMFCs) transform the chemical energy produced during a reaction between hydrogen fuel and oxygen into electrical energy. While PEMFCs are a promising source of clean energy that is self-contained and mobile, much like the alkaline fuel cells used on the U.S. space shuttle, they currently rely on expensive materials. But the substances used for catalysing these chemical reactions degrade, raising concerns about reusability and viability.
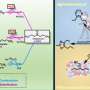
DGIST energy materials scientist Sangaraju Shanmugam and his team have developed active and durable catalysts for PEMFCs that can reduce the overall manufacturing costs. The catalysts were nitrogen-doped carbon nanorods with ceria and cobalt nanoparticles on their surfaces; essentially, carbon nanorods containing nitrogen, cobalt and ceria. Ceria (CeO2), a combination of cerium and oxygen, is a cheap and environmentally friendly semiconducting material that has excellent oxygen reduction abilities.
The fibres were made using a technique known as electrospinning, in which a high voltage is applied to a liquid droplet, forming a charged liquid jet that then dries midflight into uniform, nanosized particles. The researchers' analyses confirmed that the ceria and cobalt particles were uniformly distributed in the carbon nanorods and that the catalysts showed enhanced electricity-producing capacity.
The ceria-supported cobalt on nitrogen-doped carbon nanorod catalyst was found to be more active and durable than cobalt-only nitrogen-doped carbon nanorods and platinum/carbon. They were explored in two important types of chemical reactions for energy conversion and storage: oxygen reduction and oxygen evolution reactions.
The researchers conclude that ceria could be considered among the most promising materials for use with cobalt on nitrogen-doped carbon nanorods to produce stable catalysts with enhanced electrochemical activity in PEMFCs and related devices.
More information: Arumugam Sivanantham et al, A synergistic effect of Co and CeO2 in nitrogen-doped carbon nanostructure for the enhanced oxygen electrode activity and stability, Applied Catalysis B: Environmental (2017). DOI: 10.1016/j.apcatb.2017.08.063
Provided by DGIST (Daegu Gyeongbuk Institute of Science and Technology)