Evidence that buckyballs and carbon nanotubes form from the dust and gas of dying stars
Astronomers at the University of Arizona have developed a theory to explain the presence of the largest molecules known to exist in interstellar gas.
The team simulated the environment of dying stars and observed the formation of buckyballs (carbon atoms linked to three other carbon atoms by covalent bonds) and carbon nanotubes (rolled up sheets of single-layer carbon atoms). The findings indicate that buckyballs and carbon nanotubes can form when silicon carbide dust—known to be proximate to dying stars—releases carbon in reaction to intense heat, shockwaves and high energy particles.
"We know from infrared observations that buckyballs populate the interstellar medium," said Jacob Bernal, who led the research. "The big problem has been explaining how these massive, complex carbon molecules could possibly form in an environment saturated with hydrogen, which is what you typically have around a dying star."
Rearranging the structure of graphene (a sheet of single-layer carbon atoms) could create buckyballs and nanotubes. Building on that, the team heated silicon carbide samples to temperatures that would mimic the aura of a dying star and observed the formation of nanotubes.
"We were surprised we could make these extraordinary structures," Bernal said. "Chemically, our nanotubes are very simple, but they are extremely beautiful."
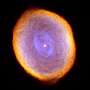
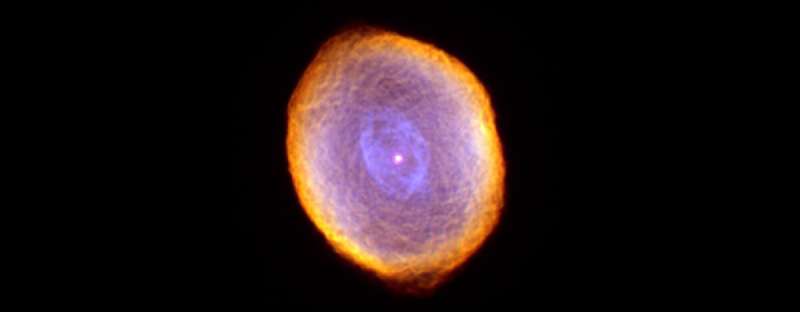
Buckyballs are the largest molecules currently known to occur in interstellar space. It is now known that buckyballs containing 60 to 70 carbon atoms are common.
"We know the raw material is there, and we know the conditions are very close to what you'd see near the envelope of a dying star," study co-author Lucy Ziurys said. "Shock waves pass through the envelope, and the temperature and pressure conditions have been shown to exist in space. We also see buckyballs in planetary nebulae—in other words, we see the beginning and the end products you would expect in our experiments."
The research was published in The Journal of Physical Chemistry A.
More information: Jacob J. Bernal et al, Destructive Processing of Silicon Carbide Grains: Experimental Insights into the Formation of Interstellar Fullerenes and Carbon Nanotubes, The Journal of Physical Chemistry A (2022). DOI: 10.1021/acs.jpca.2c01441
Provided by National Science Foundation