July 26, 2022 feature
The central core clock machinery drives the majority of metabolic rhythms
Life on earth is based on recurring 24-hour environmental cycles that are genetically encoded as molecular clocks active in all mammalian organs. Communication between these clocks can control circadian homeostasis. Temporal coordination of metabolism can then mediate inter-tissue communication. In a new report now published in Science Advances, Paul Petrus and a team of interdisciplinary researchers in epigenetics and metabolism, health sciences, computer science and biomedicine at the University of California, Irvine, U.S., and the Pompeu Fabra University in Barcelona, Spain, characterized the process to which clocks across diverse organs controlled systematic metabolic rhythms. This trajectory is a research area that hitherto remains to be explored. The team studied the metabolome of serum from mice with tissue-specific expression of the clock gene Bmal1. The experimental outcomes indicated that the central clock regulated the metabolic rhythms via behavior. The findings highlighted the circadian connection between tissues to emphasize the significance of the central clock governing the signals.
In sync with the cycle of the Earth
The Earth spins in a 24-hour cycle around its own axis and life has adapted this evolutionary trait as a genetically encoded molecular clock, referred to as the core clock machinery. Every organ in the mammalian body has a clock that cooperates to regulate circadian homeostasis. Inter-organ communication is based on metabolic fluctuations that considers the supply and demand of various tissues. The underlying basis of coordination for specific tissue clocks to regulate systematic metabolism remains a topic to be explored. In this work, Petrus and the team explored how the specific tissue clocks regulated systematic metabolism by analyzing the role of local clocks to drive the strong temporal metabolic coherence existing among serum, liver and muscle in a process taking up to 24 hours to complete and integrate to regulate circadian homeostasis.
Metabolic rhythms are linked with the core clock machinery comprising a transcriptional-translational feedback loop that's synced for 24-hours. To investigate this mechanism, the team explored time-specific Bmal1 expression in gene knockout animal models, where the genes of interest were experimentally knocked down to understand the effect of specific molecular mechanisms on the core clock and on regulating metabolism.
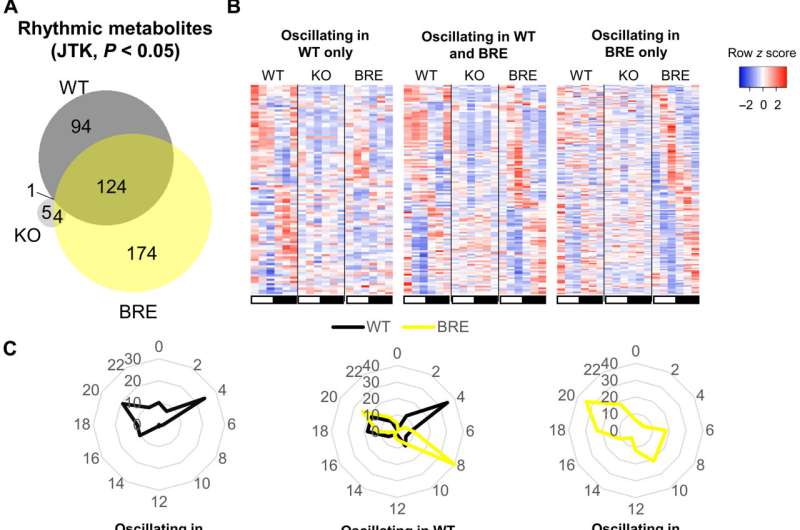
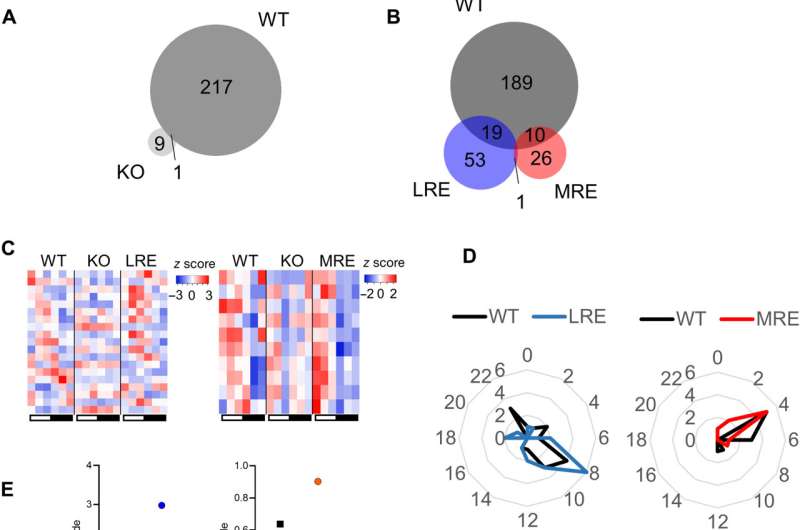
During the experiments, the researchers euthanized mice every four hours within a 24-hour diurnal cycle and collected serum from all mice cohorts. They analyzed the serum using global metabolomics via liquid chromatography mass spectrometry (LC/MS). While the wild type, also known as "normal" mice cohort, showed significant circadian oscillations detected via circulating metabolites, the Bmal1 knockout mice cohorts had lost rhythmicity of all metabolites observed in the wild type, except for cysteine-S-sulfate. The results confirmed the importance of Bimal1 expression for oscillations in circulatory metabolites. The study outcomes reaffirmed how local peripheral clocks in isolation are insufficient to drive the majority of circadian metabolic output into circulation, further highlighting the dependence of a majority of circulatory metabolites on other tissue clocks or the communication between tissue clocks.
Brain clocks
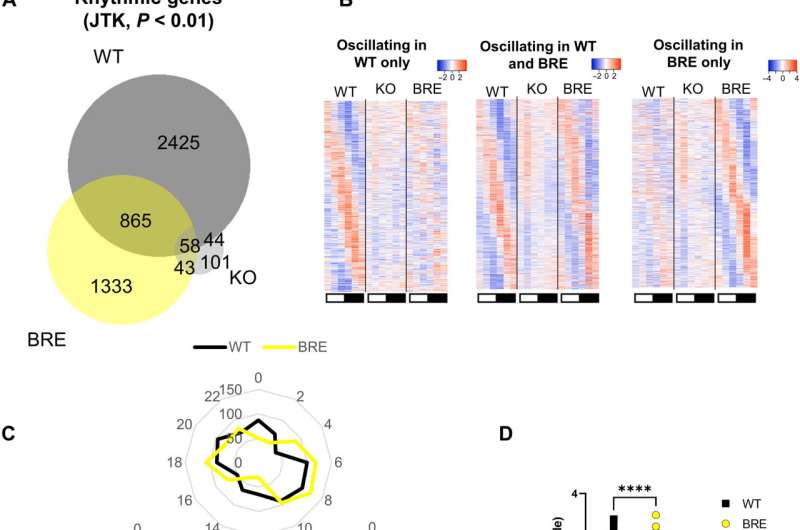
Metabolic rhythms usually thrive on food intake or from stores of energy within the body. Rhythmic feeding behavior is therefore a determinant of systemic metabolic oscillations. Behavioral rhythms are usually regulated by the central clock of the suprachiasmatic nucleus, within which the central circadian pacemaker is located and highly expresses the Syt10 gene. Knocking out Bmal1 in Syt10 expressing neurons resulted in arrhythmic behavior in mice placed in constant darkness. After further experiments, the team restored Bmal1 expression in Syt10 neurons to understand if the process restored behavioral rhythms relative to locomotor activity, metabolism and feeding behavior when compared to the wild type and knockout animal models. The scientists noticed partially rescued body weight for the mice, including total activity, food intake and adiposity as trends toward a rescue. The circadian rhythms relative to behavior and metabolism were also partially restored. The partial restoration emphasized the requirement for clocks in other cell types of the brain for full restoration of behavioral rhythms. The data also suggested the influence of the central clock to drive most circadian circulating metabolic rhythms, while the phase and amplitude required additional regulation through other clocks.
Metabolism and the core-clock
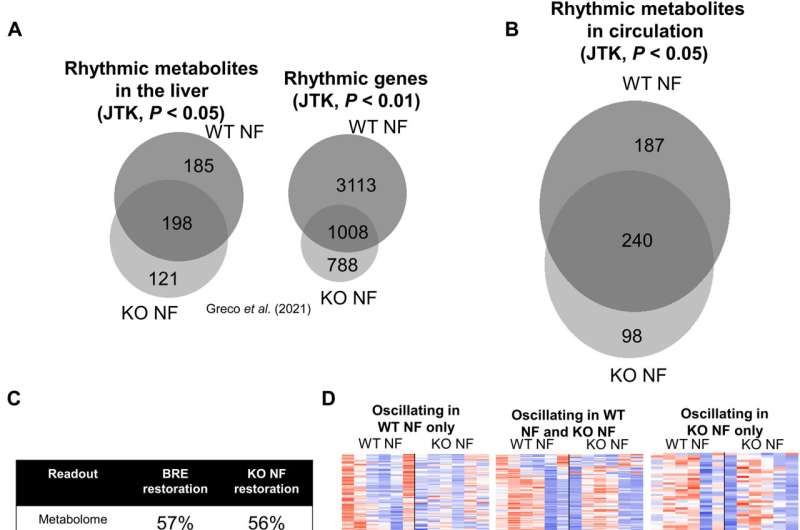
The researchers further studied the degree to which the central clock could regulate transcriptional oscillations in the absence of peripheral clocks, via RNA-sequencing studies for more than 24-hours. Some mechanisms appeared to be regulated via systemic metabolism, independent from the core clock machinery. They further showed how glucose metabolism relied on the circadian system, while systemic glucose homeostasis regulated clocks in multiple organs. The results emphasized the significance of regulating glucose homeostasis and even provided evidence to how shift work linked to diabetes. The team also studied the process by which the central clock regulated systemic metabolism. The outcomes showed how the circulating metabolic rhythm of more than 56% Bmal1-kcnockout mice could be rescued by establishing a feeding rhythm.
Outlook
In this way, Paul Petrus and colleagues dissected the complex mechanisms of circadian inter-organ communication. They showed how the central clock drove systemic rhythms to a large extent by regulating feeding-fasting rhythms. The work emphasized food as a major synchronizing factor contributing to the role of the suprachiasmatic nucleus as a master pacemaker. The work presents the central core clock as a driver of systemic metabolic rhythms. Future work will shed light on interpreting peripheral clocks to implement into clinical therapies to treat disrupted circadian rhythms.
More information: Paul Petrus et al, The central clock suffices to drive the majority of circulatory metabolic rhythms, Science Advances (2022). DOI: 10.1126/sciadv.abo2896
Kenneth A. Dyar et al, Atlas of Circadian Metabolism Reveals System-wide Coordination and Communication between Clocks, Cell (2018). DOI: 10.1016/j.cell.2018.08.042
Journal information: Science Advances , Cell
© 2022 Science X Network