How biology's hardest working pigments and 'MOFs' might just save the climate
Some of the economic sectors that are the hardest to decarbonize would benefit from the emergence of substantially more efficient catalysts involved in energy conversion chemical reactions. A breakthrough here might depend upon the use of pigments widely deployed in biological processes integrated as a catalyst into novel and highly porous molecular structures that act sort of like sponges.
A paper describing the state of play in this field and the challenges it faces was published in the journal Nano Research Energy on May 29.
In recent years, porphyrins and metalloporphyrins have played an increasingly important role in biomimetic chemistry, solar energy utilization, medicine, and a great many other applications. But use of porphyrins in electrocatalysis and photocatalysis reactions central to many energy conversion processes useful for the clean transition was found to be unstable, deactivate, and difficult to recycle, which has limited the further development of these energy conversion technologies.
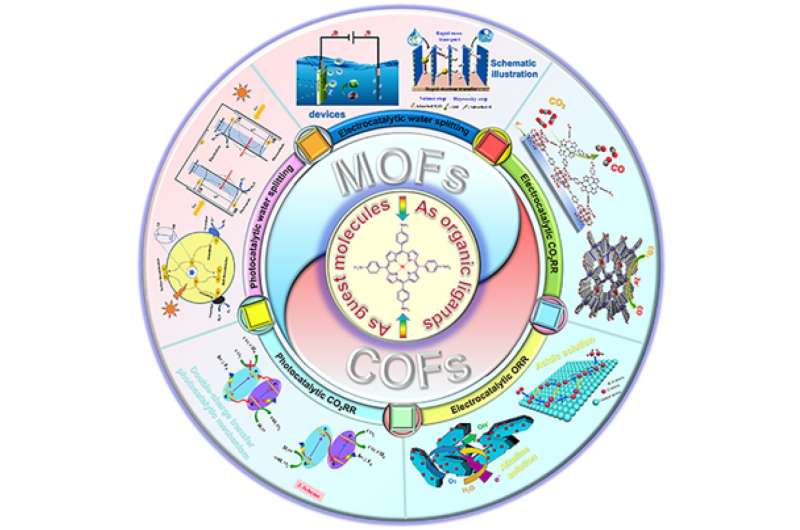
So scientists have begun to consider the integration of porphyrins as the organic ligands (the ion that binds to a central metal atom in a complex molecule) into synthetic molecular structures known as metal-organic frameworks (MOFs) and their twin, covalent-organic frameworks (COFs)—known as porphyrin-based framework materials.
"This should in principle deliver excellent electrocatalysis and photocatalysis performance as the MOF and COF structures are simple to synthesize and highly designed, thus much more controllable and structurally stable," said Yusuke Yamauchi, a co-author of the paper and researcher with the Australian Institute for Bioengineering and Nanotechnology at the University of Queensland.
"The researchers, who are themselves involved in porphyrin-based framework materials development, put together a review article describing the state of play in their field. Such review papers are necessary for young fields to advance as they clarify current understanding, discuss advances and challenges, identify research gaps and can even offer guidelines for policy and tips on best practice," Huan Pang, a co-author of the paper and the researcher with the School of Chemistry and Chemical Engineering at the Yangzhou University, China
The paper explores all the current and potential applications of porphyrin-based framework material catalysts, and finds that there remains great potential, but the field confronts several challenges.
In an economy of net-zero greenhouse gas emissions, not everything can be electrified—particularly long-haul heavy transport—and so some form of clean fuels, such as carbon-neutral synthetic hydrocarbons, ammonia or hydrogen will be necessary. All these fuels involve the conversion of clean energy—whether from the sun, wind, water or uranium—into transportable and stable chemical energy. Part of this process requires the production of clean hydrogen through the use of electricity, light or heat to split water into its constituent elements, hydrogen and oxygen.
Hydrocarbons are composed of differing ratios of carbon and hydrogen, hence the name. Thus the clean, synthetic versions replacing their dirty fossil cousins will require drawing down carbon dioxide from the atmosphere and transforming it into various usable forms of carbon as an input to be married to the clean hydrogen. To draw down atmospheric carbon and make use of it is also known as carbon capture and utilization (CCU).
All these processes, and many others involved in the clean transition (the move from fossil fuels to clean technologies) such as the use of fuel cells and light collection, are in effect chemical reactions that convert energy from one form to another, more usable form. These chemical reactions require addition of substances known as catalysts that speed the reaction up. Some of those catalysts are extremely expensive such as platinum, or are not efficient enough for the end product to compete with fossil fuels, or produce their own environmental challenges.
Thus the hunt is on for more efficient, cheaper and cleaner catalysts such as porphyrin.
The development of efficient non-precious porphyrin-based framework material catalysts to replace precious metal catalysts remains a significant hurdle. The design and construction of porphyrin blocks currently mainly relies on a highly symmetrical design, which limits the diversity of porphyrin framework families and affects their potential catalytic applications. Novel structures that employ porphyrin units with asymmetric design should be considered to extend the substance's utility.
The cost of preparing porphyrin framework materials remains high and so it is urgent that engineers develop new synthesis methods if these catalysts are to be taken up in large-scale industrial applications. Reducing the number of steps required in synthesis is an important research, but it is also extremely difficult to do this.
They conclude however that should such challenges be overcome, porphyrin-based framework materials could be a game-changer in the commercialization of energy conversion processes essential for some of the sectors that are the very hardest to decarbonize.
Porphyrins are some of biology's hardest working substances. This class of pigments is deployed in a wide array of vital processes, from photosynthesis to breathing. Derivatives of these water-soluble, ring-shaped molecules that bind metal ions include chlorophylls in plants and the hemoglobins that carry oxygen in the blood of animals. They also enhance the catalytic activities of enzymes in a range of other life-giving chemical reactions. Metalloporphyrins are of particular interest with respect to the clean transition due to their role as catalysts in water splitting to produce hydrogen and oxygen.
More information: Jiawei Gu et al, Porphyrin-based framework materials for energy conversion, Nano Research Energy (2022). DOI: 10.26599/NRE.2022.9120009
Provided by Tsinghua University Press