Nano-protein corona perturbs protein homeostasis and remodels cell metabolism
A research team led by Prof. Chen Chunying from the National Center for Nanoscience and Technology (NCNST) of the Chinese Academy of Sciences (CAS) has recently investigated the evolution of the nano-protein corona during endocytosis and its disturbance to protein homeostasis and cell metabolism. Their results were published in PNAS.
When nanoparticles enter biological systems, biomolecules of biological fluid quickly bind to the surface of the nanoparticles. The nano-protein corona formed by interacting with protein molecules in the blood as a starting step has a huge impact on the transport and fate of the nanoparticles. How the formation of the nano-protein corona affects the recognition, transport, distribution, function and biological effects of nanoparticles in the tissues and cells of different barrier systems is a "black box" for medical application of nanomaterials, which not only restricts the delivery efficiency of nanomedicine, but also seriously affects effectiveness and safety.
An important challenge in this area is the complexity of the nano-protein corona, which is influenced by the diversity of biomolecules in different tissues and organs, as well as physiological and pathological states. At present, there is urgent need to understand how the protein composition and structural characteristics of the protein corona evolve within biological microenvironments.
To solve this problem, the researchers have revealed the dynamic evolution pattern of the protein composition of the nano-protein corona in the process of cell transport through the innovative application of multi-dimensional multi-omics (proteomics, metabolomics, lipidomics), intermolecular interactions, and in situ mass spectrometry imaging.
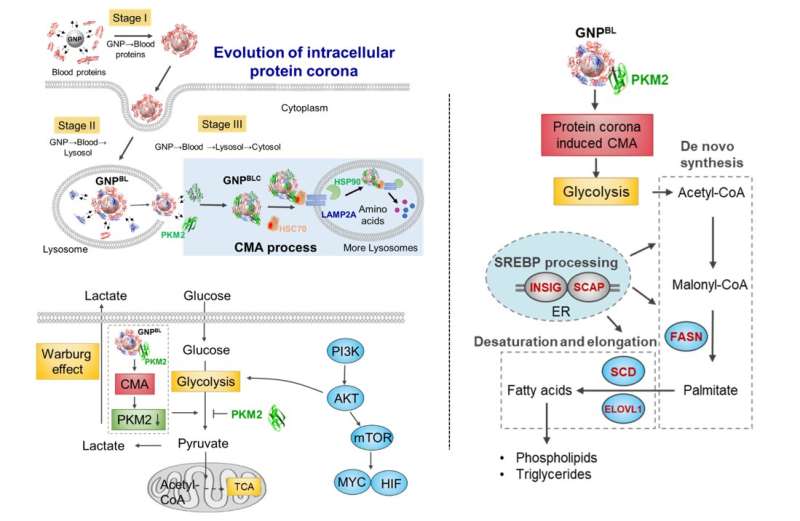
Taking gold nanoparticles as the model, the dynamic evolution process of the protein corona from the blood system to the intracellular (blood-lysosomal-cytoplasm) was studied. When the nanoparticles were endocytosed into the lysosome from the blood environment, and then escaped from the lysosome into the cytoplasm, the protein composition on the surface of the nanoparticles would change dramatically. Most were replaced by intracellular protein molecules, retaining only part of the protein corona components formed in the blood environment.
Subsequently, intracellular evolution of the nano-protein corona not only disrupted intracellular protein homeostasis (proteostasis), but also triggered the enrichment of chaperone proteins (HSC70, HSP90) and pyruvate kinase M2 (PKM2) on the surface of the intracellular nano-corona, and stimulated chaperone-mediated autophagy. It further affected cell glycolysis, causing changes to the cell energy metabolism and regulating cell lipid metabolism process.
This study elucidates the evolutionary pattern of nanoparticles from blood to a subcellular microenvironment, and identifies the specificity of the intracellular microenvironment of nano-protein corona, thereby reshaping cell metabolism. It also provides theoretical support for an in-depth understanding of the complex biological effects of nanomaterials and nanobiotic interface regulation.
More information: Rong Cai et al, Dynamic intracellular exchange of nanomaterials' protein corona perturbs proteostasis and remodels cell metabolism, Proceedings of the National Academy of Sciences (2022). DOI: 10.1073/pnas.2200363119
Journal information: Proceedings of the National Academy of Sciences
Provided by Chinese Academy of Sciences