Exploring new roles for non-coding RNA
The past decade has seen enormous advances in the understanding of regulatory noncoding RNAs, according to a Northwestern Medicine review published in Molecular Cell.
In particular, continued study of downstream-of-gene (DoG) transcripts could reveal the mechanisms that promote RNA processing, according to Shannon Lauberth, Ph.D., associate professor of Biochemistry and Molecular Genetics and senior author of the review.
"The discovery of DoGs has laid the foundation for the next decade to be an exciting time for unveiling the many enigmas that control DoG biogenesis and function," Lauberth said.
The majority of DNA in the human genome does not code for proteins, instead creating noncoding RNAs (ncRNAs). These ncRNAs regulate gene expression, sometimes through the RNA polymerase II (RNAPII) transcription complex, which transcribes DNA into messenger RNA (mRNA) as an intermediate step in creating proteins.
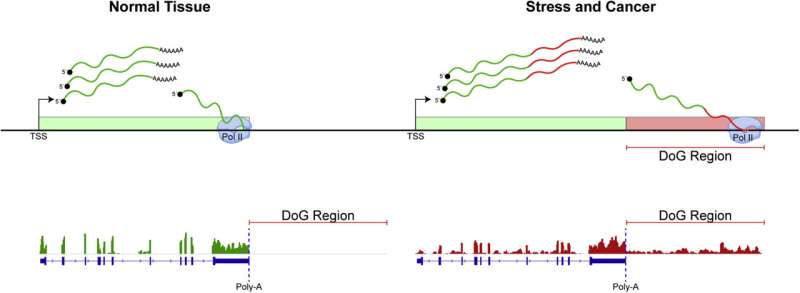
One important class of ncRNA are DoG transcripts, located just downstream of the gene boundary of protein-coding genes. DoGs are increasingly identified as hallmarks of cellular stress and disease, according to Lauberth.
Recent studies have shown that errors preventing RNAPII from properly terminating at the ends of genes leads to transcription "read through." Termination ensures the transcriptional machinery comes to a halt at the ends of protein-coding genes, and failed termination leads to subsequent expression of DoGs.
Failed termination and DoG expression can lead to invasive transcription, a process where downstream genes are aberrantly expressed due to the lack of a genetic "red light" at the end of the upstream gene.
However, new tools for identifying "read through" transcription are emerging, and future study of DoGs is a promising area of study in molecular genetics, Lauberth said.
"Research studies focused on ncRNAs is bringing about a paradigm shift in how the scientific community views the noncoding genome and RNA-mediated gene regulation," Lauberth said. "Raising awareness of the emerging classes of ncRNAs, including DoGs and their associated phenotypes, is an issue of great significance in light of the emerging field of functional DoGs and the potential of using DoGs as diagnostic markers and therapeutic targets for the tuning of gene expression programs that shift cellular stress and disease-related networks."
More information: Marc Morgan et al, It's a DoG-eat-DoG world—altered transcriptional mechanisms drive downstream-of-gene (DoG) transcript production, Molecular Cell (2022). DOI: 10.1016/j.molcel.2022.04.008
Journal information: Molecular Cell
Provided by Northwestern University