Heart progenitors spontaneously regenerate cardiac muscle via a tight junction 'honeycomb' in salamanders

Whether there are endogenous adult heart progenitors that can replenish damaged muscle cells remained controversial. Now researchers at Karolinska Institute in Sweden show that the outermost layer of the heart, called epicardium, acts as a source of cardiac muscle cells through formation of an intriguing honeycomb-like structure.
The study, published in Nature Cell Biology, identifies a new paradigm for heart regeneration by studying a salamander model for heart regeneration and reports an important role for tight junctions in the process.
Heart disease remains the number one killer worldwide, and human hearts have a very limited regenerative capacity. In contrast, teleost fish and amphibians can regenerate their hearts. This has been mainly attributed to the dedifferentiation and proliferation of the remaining muscle cells, which does not readily happen in humans.
A team of researchers in the Department of Cell and Molecular Biology now show that there is an alternative strategy to regenerate the heart muscle which may lead to new therapeutic strategies. "Salamanders have amazing regenerative features and we have during the recent years developed tools and methods to better understand the underlying cellular and molecular mechanisms. These tools have been instrumental in exploring how salamanders create new heart muscle," says Professor András Simon who co-supervised the study.
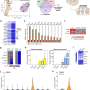
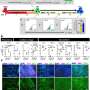
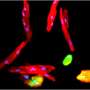
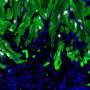
By developing a genetic marker independent lineage tracing system in transgenic reporter salamanders, researchers were able to show continuous epicardial contribution to heart muscle cell, which was greatly expanded in response to injury. Through single cell RNA sequencing researchers identified expression of the tight junction gene Claudin-6 in the salamander epicardium. After the injury epicardial cells expanded and migrated to the injury site and formed a honeycomb-like structure decorated by Claudin-6 protein. "To study the importance of this structure we engineered a naturally occurring toxin that can recognize and break it. The results were remarkable, as in the absence of this three-dimensional organization salamander hearts did not regenerate. To our knowledge this is the first time such collective cell behavior is observed in cells derived from the epicardium in response to injury," added first author Elif Eroglu, a senior scientist in the Chien Lab, who led the study.
Similar epicardial cells also exist in the human heart. "This work establishes a new cellular and molecular paradigm for adult heart muscle regeneration, and could offer new hope to millions of patients waiting for a heart transplant," says Distinguished VR Professor Ken Chien who co-supervised the study.
More information: Elif Eroglu et al, Epicardium-derived cells organize through tight junctions to replenish cardiac muscle in salamanders, Nature Cell Biology (2022). DOI: 10.1038/s41556-022-00902-2
Journal information: Nature Cell Biology
Provided by Karolinska Institutet