Controlling chemical mirror images
Chirality, while not a rarity in the world of molecules, is nevertheless a special property. If a molecule is chiral (from the Greek word chiros = hand), it exists in two mirrored versions that are very similar but not identical—like two hands that can be folded together, but cannot be placed congruently on top of each other. This is why we speak of right-handed and left-handed molecules, or enantiomers, which means "opposite shape" in Greek.
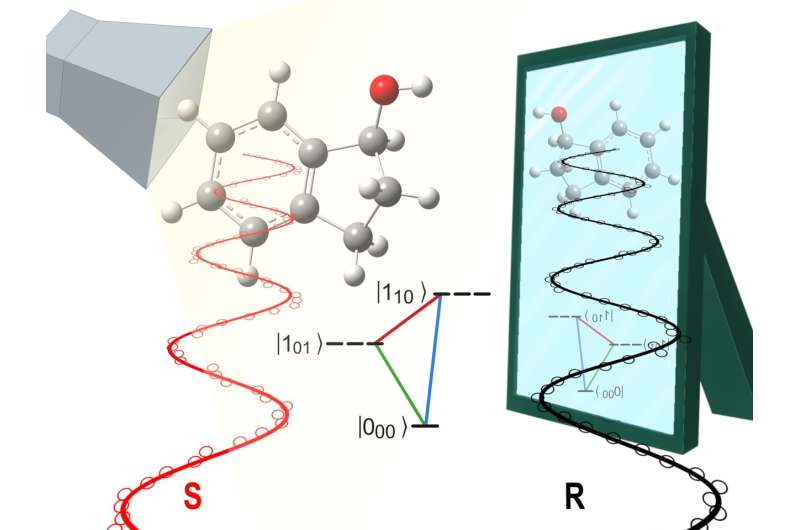
An international team of scientists from the Fritz Haber Institute of the Max Planck Society and the Prokhorov General Physics Institute of the Russian Academy of Sciences has found a way to address these molecules separately. Since chiral molecules are very similar to each other, this is a real challenge. "The trick is to expose them to electromagnetic radiation in a way so that only one 'hand,' i.e., one enantiomer, responds. This allows us to specifically control right- or left-handed molecules and learn more about them," says Dr. Sandra Eibenberger-Arias, head of the Controlled Molecules group at the Fritz-Haber-Institut.
Learning this is important because enantiomers sometimes have very different biological and chemical qualities, for which explanations are sought. Take, for example, the chiral molecule carvone: one "hand" smells like mint, the other like caraway. Or the notorious sedative thalidomide, which is named after its active ingredient, a chiral molecule: while one form had the intended sedative effect, the other caused birth defects. Eibenberger-Arias' group studies the physical properties of chiral molecules. "Theory predicts a small energy difference between the two enantiomers, due to what is called parity violation. However, this has not been shown experimentally so far," explains JuHyeon Lee of the Fritz-Haber-Institut, first author of the published results, which appeared in the journal Physical Review Letters.
With a clever combination of different methods, however, the group of scientists has come a little closer to achieving this. They irradiate chiral molecules in the gas phase with UV radiation and microwaves. As a result, right-handed and left-handed molecules are put into different rotational states by changing the microwave radiation. The researchers have thus gained more control than ever before over which "hand" is in which rotational state. They have also, for the first time, compared experimental results with accurate predictions from theory, leading to an improved understanding of the underlying physical effects.
While complete separation of the enantiomers may not yet be achieved using this method, it is remarkable that they could be controlled so successfully in the first place. This contradicts the often-used over-simplified account that they have the same physical properties. "If that were the case, we would not be able to control the enantiomers using physical methods," says Sandra Eibenberger-Arias. The international team of three female and three male scientists has thus laid a good foundation for follow-up experiments, and perhaps even for experimental proof of parity violation. This would be a milestone for basic research—and for all future applications as well.
More information: JuHyeon Lee et al, Quantitative Study of Enantiomer-Specific State Transfer, Physical Review Letters (2022). DOI: 10.1103/PhysRevLett.128.173001
Journal information: Physical Review Letters
Provided by Fritz Haber Institute of the Max Planck Society