Seeing lipids more deeply with mass spectrometry
The development of new scientific ways to see more deeply into the building blocks of nature on a cellular level has led to the some of the greatest advances in medicine over the last century. Now, new research into phosphoinositides, which are a family of membrane lipids essential for many biological and pathological processes and which represent one of the most functionally versatile membrane lipid families involved in human health and disease, has seen further developments in the use of mass spectrometry in continuing to push back the barriers leading to new treatments for many diseases.
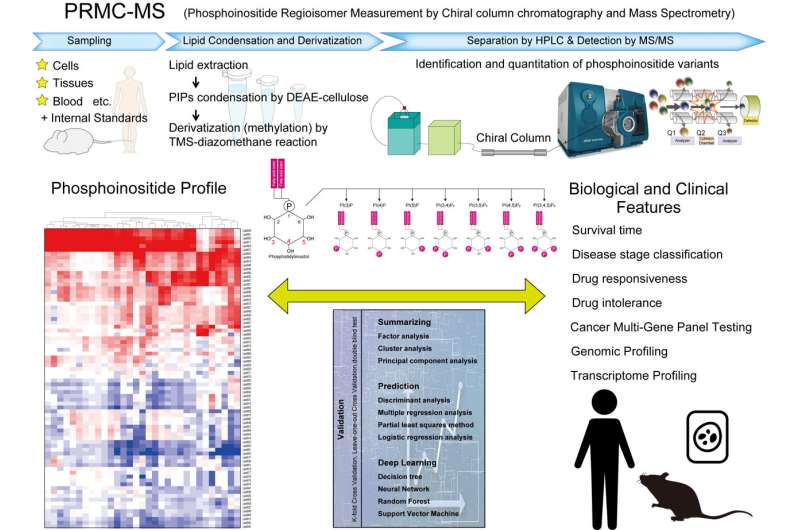
In the past, due to various reasons related to their complexity and their low intracellular concentrations, the profiling of these lipids and the linking of a specific acyl variant to biological change has been difficult. However, a new system called PRMC-MS (Phosphoinositide Regioisomer Measurement by Chiral column chromatography and Mass Spectrometry) has now enabled the characterization of the dynamics of phosphoinositide acyl variants both in intracellular and extracellular environments.
Previous methods of measuring and profiling phosphoinositides have produced results that cannot be easily applied to clinical or pathological samples from experimental animals. Even newer methods involving the use of mass-spectrometry, which have made advances in some areas, still reflect the problem of how to simultaneously quantify the acyl variants of individual regioisomers in biological samples.
The PRMC-MS method now solves this problem and points the way to an understanding of how these lipids influence cell functions. Using PRMC-MS, it is now possible to simultaneously measure all eight classes of phosphoinositides in a single sample. The highly sensitive nature of PRMC-MS allows for the detection of tiny but important changes in intracellular phosphoinositide levels, yielding data that shows that it can be applied to blood samples to track phosphoinositide signatures potentially related to disease states.
PRMC-MS enables the comprehensive analysis of phosphoinositide acyl variants in various types of biological samples, including surgical specimens, which can be used to throw a light on previously unrecognized disturbances of phosphoinositide fatty acyl profiles in cancerous tissue and to monitor their extracellular mobilization. Further study of the differing acyl variants and their conferring of protein binding properties could possibly also reveal how they activate a signaling pathway that favors cancer cell growth and survival and emerge as a target for cancer therapy. Thus, PRMC-MS may well illuminate the role played by phosphoinositides in the pathogenesis of cancers and inflammatory diseases.
In addition, the use of PRMC-MS in the evaluation of phosphoinositide signatures at the acyl variant level in tissue and liquid biopsies may reveal biomarkers suitable for a wide variety of clinical applications.
In the future, applications such as the above may greatly facilitate drug development strategies based on the devising of a therapeutic agent that pinpoints a specific pathogenic phosphoinositide acyl variant, and thus open the way for much more accurate therapeutic methods and cures for patients suffering from a range of diseases that have proven difficult in the past.
The PRMC-MS method is covered in greater detail in a paper recently published in Nature Communications.
More information: Shin Morioka et al, A mass spectrometric method for in-depth profiling of phosphoinositide regioisomers and their disease-associated regulation, Nature Communications (2022). DOI: 10.1038/s41467-021-27648-z
Journal information: Nature Communications
Provided by Tokyo Medical and Dental University