Reassessing radon as a reliable groundwater tracer
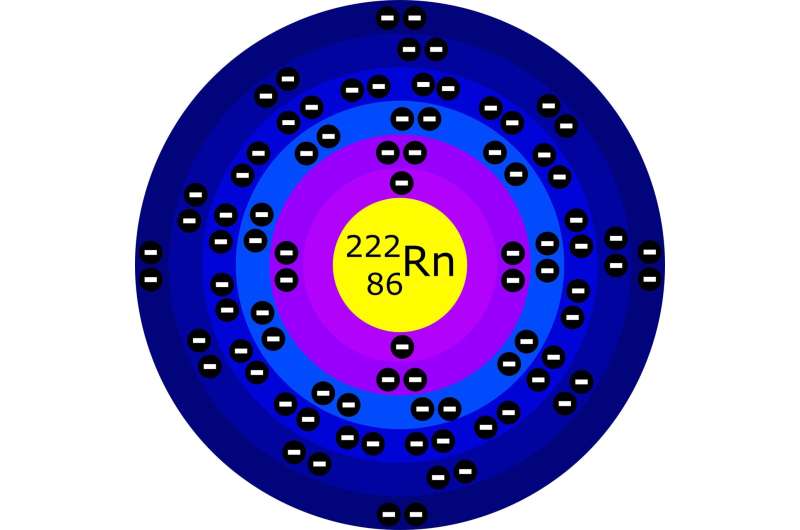
All radioactive materials which naturally occur in water will produce at least one isotope of radon as a decay product. As radioactive atoms are transported through groundwater aquifers in the form of gaseous radon, they are eventually transferred to the atmosphere. Measurements of the rate of this transfer can, in theory, be used to trace the infiltration of water into the surrounding soil. Yet in a new study detailed in EPJ Plus, researchers in Egypt and Saudi Arabia, led by Elsayed Elmaghraby at the Egyptian Atomic Energy Authority, show that this technique could have a significant flaw.
Currently, researchers use the transfer of radon from groundwater to assess factors ranging from the environmental impacts of ore extraction to earthquake precursors. The overall rate at which radon is transferred to the atmosphere is governed by several processes: including its formation through radioactive decay; transport to the water's surface; adsorption onto other surrounding atoms; and diffusion under concentration gradients. In measuring this rate, researchers typically assume that radon's concentration is in equilibrium with its own radioactive decay products.
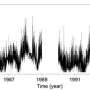
To test this assumption, Elmaghraby's team prepared a water-based solution containing thorium and uranium, and then left it in isolation for two years. Afterwards, they exposed the solution to the atmosphere, and measured how the resulting radon transfer rate evolved over time. Subsequently, they simulated the experiment with a mathematical model, accounting for all relevant processes.
These simulations revealed that during the liquid's isolation, its decay products had partially re-dissolved in the water to varying degrees, depending partially on the geometry of the medium. This can vary widely in the presence of grains like sand or soil, which come in a diverse array of shapes and sizes. In addition, re-dissolving was affected by the presence of bubbles, containing gasses such as water vapor and helium—another natural decay product. Ultimately, this meant that radon and its decay products cannot be in equilibrium, throwing doubt on previous measurements of the overall radon transfer rate.
More information: Elsayed K. Elmaghraby et al, Radon exhalation and transfer processes in aqueous media, The European Physical Journal Plus (2021). DOI: 10.1140/epjp/s13360-021-02231-z
Provided by Springer