One-step strategy to synthesize site-specific antibody-drug conjugates with LacNAc-based substrates
Antibody-drug conjugates (ADCs), a complex covalently linking a potent cytotoxin onto the antibody, has attracted great interest as a novel biotherapeutic approach. Site-specific conjugation of potent payloads can improve the therapeutic index of ADCs compared with random conjugation. Glycosite-specific ADCs (gsADCs), harnessing Asn297 N-glycan of IgG Fc as the conjugation site for drug payloads, usually require multi-steps for IgG glycoengineering with two or more enzymes, thus limiting the substrates diversification and complicating the preparation process.
To solve these problems, a research team led by Huang Wei from the Shanghai Institute of Materia Medica of the Chinese Academy of Sciences developed a facile and highly efficient method for synthesizing gsADCs in one-step with a disaccharide-drug substrate. The study was recently published in Acta Pharmaceutica Sinica B.
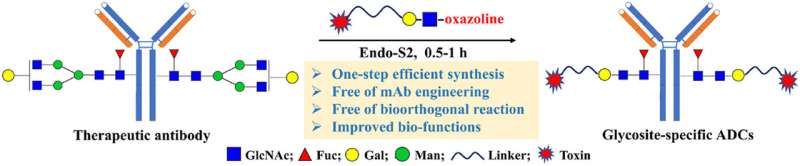
The researchers firstly screened a series of glycan substrates and ENGases for one-step IgG glycoengineering. They found that N-acetyl-lactosamine (LacNAc) could be transferred onto the N-glycosylation site of native Trastuzumab in one step without hydrolysis, and the derived LacNAc substrates with various functional groups and small molecules also showed great transglycosylation activity. The glycoengineered Trastuzumabs equipped with bioorthogonal groups were further applied for Fc receptors binding determination, fluroscence labeling and glycan-elongation by click chemistry.
Encouraged by the robust LacNAc-based IgG glycoengineering, the researchers applied this method in one-step or two-step synthesis of glycosite-specific ADCs. First, the obtained LacNAc-based Trastuzumab with an azido tag can readily react with a cyclooctyne-tagged payload to give a gsADC in two-step via click chemistry. More importantly, the researchers prepared a MMAE-LacNAc substrate and found that the complex could be recognized by Endo-S2 and transferred onto the glycosylation site within one hour to give the corresponding gsADC in one-step. The obtained gsADCs with truncated glycan LacNAc showed excellent homogeneity, stability and improved anti-tumor activity.
This study presents a novel strategy for highly efficient synthesis of gsADCs by reprograming the multi-step IgG glycoengineering to a one-step manner with LacNAc-based substrates. The strategy is also compatible with other antibody types, such as bispecific antibodies and nanobodies with Fc domain.
More information: Wei Shi et al, One-step synthesis of site-specific antibody–drug conjugates by reprograming IgG glycoengineering with LacNAc-based substrates, Acta Pharmaceutica Sinica B (2021). DOI: 10.1016/j.apsb.2021.12.013
Provided by Chinese Academy of Sciences