New fluorescent nanovesicles for intracellular biomarker detection
A new work by the Nanomol Group, belonging to the CIBER-BBN network, together with a team from the University of Rome Tor Vergata, presents new nanovesicles capable of crossing biological barriers such as cell membranes, while maintaining their sensing capacity, which makes them attractive probes for intracellular detection of biomarkers.
"The development of probes capable of sensing the biological environment and signaling the presence of a specific target molecule is a challenge with relevance in a variety of biomedical applications, from drug delivery to diagnostic tools," says Mariana Köber, ICMAB researcher and corresponding author of the study, together with Nora Ventosa, from ICMAB, and Alessandro Porchetta, from the University of Rome Tor Vergata.
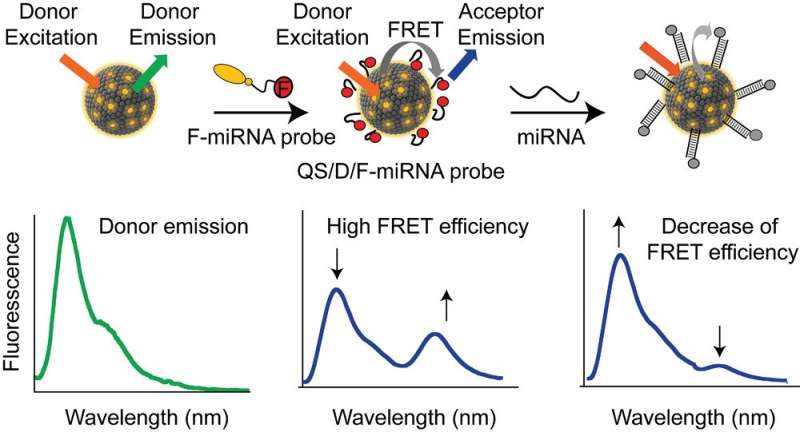
This work, which has been published in Advanced Functional Materials, presents the design of fluorescent nanovesicles functionalized with biomimetic DNA capable of translating their binding to a target molecule into an optical output, through a change in Förster resonance energy transfer (FRET) and fluorescent emission.
These Quatsomes (QS) nanovesicles are an emerging class of highly stable small unilamellar vesicles of ≈50-100 nm in diameter, formed by the self-assembly of ionic surfactants and sterols in aqueous media. Their high stability, also in body fluids, unilaminarity and particle-to-particle homogeneity make them an attractive soft material for sensing applications. "QS nanovesicles are loaded with fluorescent probes based on amphiphilic nucleic acids to produce programmable FRET active nanovesicles that function as highly sensitive signal transducers," explain the researchers.
The CIBER-BBN researchers have participated in the characterization of the photophysical properties of these nanovesicles and have demonstrated highly selective detection of clinically relevant microRNAs with sensitivity in the nanomolar range. This production of the nanovesicles and their physicochemical characterization has been carried out thanks to the services of the ICTS NANBIOSIS, through the Biomaterials Processing and Nanostructuring Unit of ICMAB-CSIC.
According to the authors, the proposed strategy could be easily adapted to the detection of different biomarkers: "we hope to achieve a bioimaging platform for the detection of a wide range of nucleic acids and other clinically relevant molecules in body fluids or directly in cells, thanks to the ability of Quatsomes for intracellular delivery."
More information: Marianna Rossetti et al, Engineering DNA‐Grafted Quatsomes as Stable Nucleic Acid‐Responsive Fluorescent Nanovesicles, Advanced Functional Materials (2021). DOI: 10.1002/adfm.202103511
Journal information: Advanced Functional Materials
Provided by Institut de Ciència de Materials de Barcelona