New tool reveals genetic influence of some sex-biased diseases, including lupus
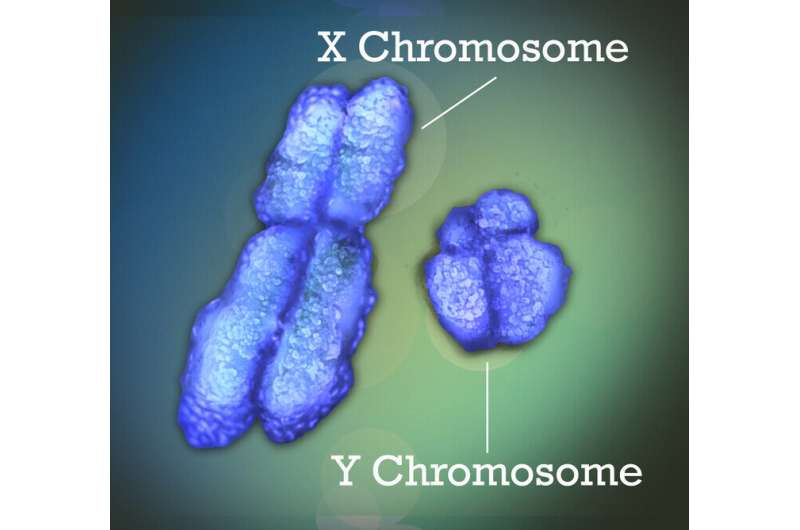
Many human diseases can differ between males and females in their prevalence, manifestation, severity or age of onset. Examples include Lupus, where more than 80% of patients are females; Alzheimer's disease, where females have higher incidence and tend to suffer quicker cognitive decline; and COVID-19 infections that are frequently more severe in males.
These sex differences may have a genetic basis that is attributable to the sex chromosomes. The X chromosome—one of the two sex chromosomes—is known to play an important role in human development and disease. New research led by Penn State College of Medicine reveals for the first time that sex-biased diseases can be attributable to genes that escape X chromosome inactivation (XCI), a process that ensures that females do not overexpress genes on their X-chromosomes.
The team developed a genetic tool that can identify these XCI escape genes, and it may also help in determining whether a female will develop a sex-biased disease and if the disease will become progressively worse over time. The tool may even be useful in understanding the sex differences in immune responses to COVID-19, as the disease is thought to produce more severe symptoms and higher mortality in men than in women.
"The X chromosome plays an important role in human development and disease, yet the X chromosome is frequently ignored in human genetic studies because of bioinformatics challenges in the analysis of the data," said Laura Carrel, associate professor of biochemistry and molecular biology, Penn State College of Medicine. "Our new method gets around these challenges and allows us to identify XCI escape genes and assess their role in sex-biased diseases. With further research and fine-tuning, we think it could serve as a predictive tool in these disorders and could lead to the identification of new disease treatments and interventions."
The human genome is organized into 23 pairs of chromosomes, one pair of which is the sex chromosomes. This pair comprises two X chromosomes for females and one X and one Y chromosome for males. Early in embryonic development in females, one of the two X chromosomes is randomly inactivated to ensure that, like in males, only one functional copy of the X chromosome—either the one inherited from the female's mother or the one inherited from her father—occurs in each cell.
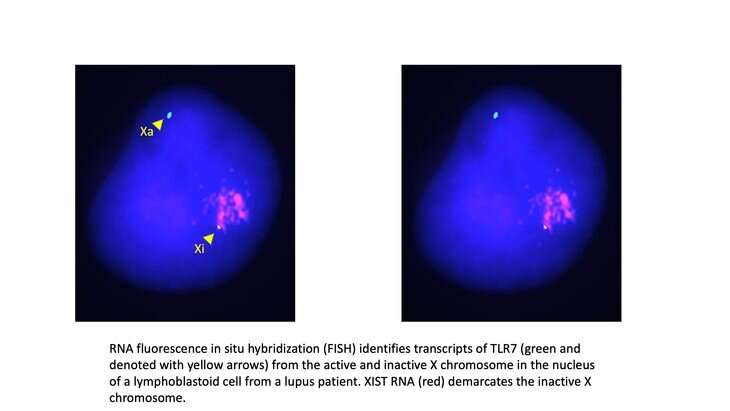
"In females, about 30% of the genes on the X chromosome escape this inactivation—or XCI—leaving them with two functional copies of those genes," said Carrel. "The question is, does having two copies of those genes make a female more susceptible to traits, such as lupus, that show a sex bias?"
To answer this question, a critical first step is to identify the XCI escape genes. Yet, conducting a chromosome-wide analysis is difficult due to the random nature of XCI in early development, as XCI affects the X chromosome that a female inherits from one parent in some cells, but the other X in other cells.
In their study, which published on Aug. 23 in the journal Genome Research, the researchers developed a novel statistical model, called XCIR (X-Chromosome Inactivation for RNA-seq), that can identify XCI escape genes using bulk RNA-sequencing data, a type of genetic data. The method separately evaluates how much a gene is expressed from each X chromosome. A gene is deemed to escape XCI if the ratio of its expression from the two X chromosomes differs significantly from genes that are known to be X inactivated. The method outperforms other approaches because it can more effectively handle the errors arising from next-generation sequencing technologies and the complex biology of XCI.
"Our method—available in an intuitive, well-documented and freely available software—is more powerful than alternative approaches and is computationally efficient to handle large population-scale datasets," said Dajiang Liu, associate professor of public health sciences and biochemistry and molecular biology, Penn State College of Medicine.
The team applied its method to a dataset including nearly half a million people, and identified hundreds of traits, including male- or female-biased diseases such as lupus, that may be influenced by these genes that escape XCI. As shown by others, the escape genes also contribute to Alzheimer's disease and response to COVID-19 infections as well.
"We have developed the methodology needed to establish XCI status for population-sized datasets," said Liu. "This work highlights the increased importance of XCI escape genes to female-biased diseases and may one day be used to accurately predict disease. Importantly, a better understanding of the sex chromosomes will be an important step in resolving health disparities between the sexes."
More information: Renan Sauteraud et al, Inferring genes that escape X-Chromosome inactivation reveals important contribution of variable escape genes to sex-biased diseases, Genome Research (2021). DOI: 10.1101/gr.275677.121
Journal information: Genome Research
Provided by Pennsylvania State University