How low did it go? Study seeks to settle debate about oxygen in Earth's early atmosphere

Scientists have long debated how much molecular oxygen was in Earth's early atmosphere. About 2.4 billion years ago, there was a rise in oxygen that transformed Earth's atmosphere and biosphere, eventually making life like ours possible. This transition is called the "Great Oxidation Event." But how much oxygen was in the atmosphere before this time?
A team of scientists, led by former Arizona State University doctoral student Aleisha Johnson, has been working to unravel the mystery of how the stage was set for the Great Oxidation Event.
Using computer modeling, Johnson and her team determined how much oxygen might have been present at Earth's surface before the Great Oxidation Event—and the implications for life on early Earth.
"We all breathe oxygen, and we all live on the only planet known where that is possible," says Johnson. "With our study, we're one step closer to understanding how that happened—how Earth was able to transition to, and sustain, an oxygen-rich atmosphere."
The results of their study have been published in Science Advances.
The long-standing puzzle
Geoscientists studying the rock record of Earth have found seemingly conflicting evidence about Earth's early atmosphere. On the one hand, the "fingerprints" of oxygen found after the Great Oxidation Event are mostly missing before that time, leading some scientists to argue that it was absent.
But recent discoveries suggest at least some breakdown of common minerals that react vigorously in the presence of oxygen, and at least some supply to the oceans of chemical elements like molybdenum that accumulate in rivers and oceans when oxygen is present. The conflicting lines of evidence create a long-standing puzzle.
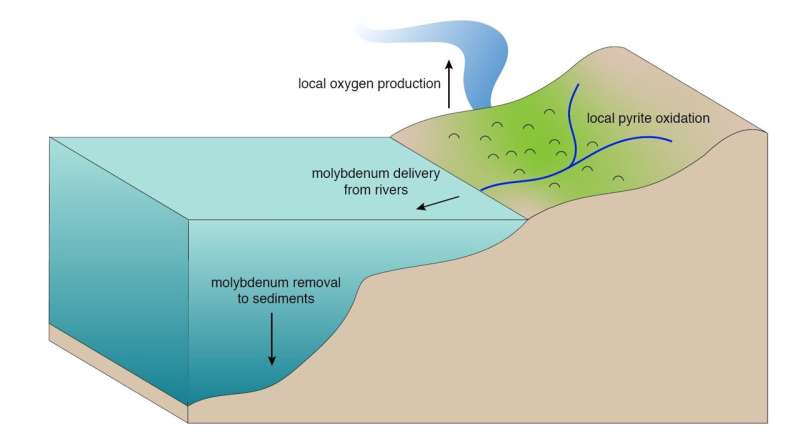
"The evidence seemed contradictory, but we knew there must be an explanation," said Johnson, who is currently a National Science Foundation postdoctoral fellow at the University of Chicago.
To help resolve this puzzle, Johnson and her team wrote a computer model that uses what is known about the environmental chemistry of molybdenum, the reactions of minerals with small amounts of oxygen, and measurements others have made of molybdenum abundances in ancient sedimentary rocks, to figure out the range of oxygen levels that was possible in Earth's atmosphere before 2.4 billion years ago.
"This computer model helps us quantify how much oxygen is actually needed to produce the chemistry that is visible in the rock record," said Johnson.
What the team found was that the amount of oxygen needed to explain the molybdenum evidence was so small that it wouldn't have left many other fingerprints.
"There's an old saying that 'absence of evidence is not evidence of absence,'" said study co-author Ariel Anbar, who is a professor at ASU's School of Earth and Space Exploration and School of Molecular Sciences. "Until now, our ideas about oxygen being absent before the Great Oxidation Event were mostly shaped by an absence of evidence. Now we have reason to think it was there—just at lower levels than could be detected before."
The findings support other lines of evidence suggesting that oxygen was being produced, possibly by biology, long before the Great Oxidation Event. That, in turn, helps scientists in their quest to figure out what changes in the Earth's systems caused one of the most important transformations in Earth's history.
"Our hope is that these constraints on ancient atmospheric oxygen help us understand the cause and nature of the Great Oxidation Event. But this isn't just about Earth history. As we begin to explore Earth-like worlds orbiting other stars, we want to know if oxygen-rich atmospheres like ours are likely to be common or rare. So this research also helps inform the search for life on planets other than our own," said Johnson.
The additional authors on this study are Chadlin Ostrander of Woods Hole Oceanographic Institution, Stephen Romaniello of the University of Tennessee, Christopher Reinhard of the Georgia Institute of Technology, Allison Greaney of Oak Ridge National Laboratory and Timothy Lyons of the University of California, Riverside.
More information: Aleisha C. Johnson et al, Reconciling evidence of oxidative weathering and atmospheric anoxia on Archean Earth, Science Advances (2021). DOI: 10.1126/sciadv.abj0108
Journal information: Science Advances
Provided by Arizona State University


















