Humans will always have oxygen to breathe, but we can't say the same for ocean life
There is nothing more fundamental to humans than the availability of oxygen. We give little thought to the oxygen we need, we just breathe, but where does it come from?
To shed light on this, statements such as "the ocean provides 50% of the oxygen we breathe," or its equivalent, "every second breath we breathe comes from the ocean," have become common mantras to highlight human dependence on the ocean and the risk of lower oxygen supply due to climate change and environmental degradation.
These mantras are repeated by high-profile politicians, including US climate envoy John Kerry and French president Emmanuel Macron, international organizations such as Unesco and the European Commission, and even prominent reports from the IPCC and other reputable scientific institutions.
While they may be good fodder for speeches, these claims misrepresent where the oxygen we breathe actually comes from, and in doing so, mislead the public as to why we should step up our role as ocean custodians.
Where do we get our oxygen?
The Earth's atmosphere has not always been as rich in oxygen as it is today. The atmosphere is now made up of 21% oxygen, but it accounted for just 0.001% of today's levels during the first 2 billion years of Earth's history.
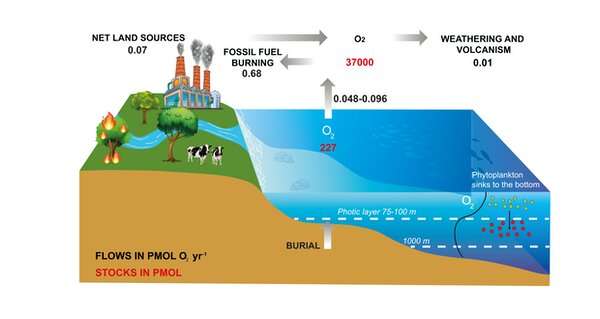
It is the advent of microscopic ocean bacteria and plants (phytoplankton) and, later, larger plants on land which caused the staggering increase of oxygen in our atmosphere. This oxygen is derived from photosynthesis—the process by which plants turn carbon dioxide and water into organic matter and oxygen.
Oxygen has been relatively stable at a high level for the past 500 million years. Today, roughly half of photosynthesis takes place in the ocean and half on land.
So yes, the ocean is responsible for about 50% of the oxygen produced on the planet. But it's not responsible for 50% of the air we humans breathe. Most of the oxygen produced by the ocean is directly consumed by the microbes and animals that live there, or as plant and animal products fall to the seafloor. In fact, the net production of oxygen in the ocean is close to 0.
A tiny fraction of the primary production, roughly 0.1%, escapes degradation and is stored as organic carbon in marine sediments—a process referred to as the biological carbon pump. This organic carbon may eventually turn into fossil fuels such as coal, oil and gas. The tiny amount of oxygen which had been generated to produce this carbon store can later be released to the atmosphere. A similar process occurs on land too, with some carbon stored in soils.
Therefore, the oxygen we currently breathe comes from the slow accumulation of O2 in the atmosphere supported by the burial of organic matter over very long time-scales—hundreds of millions of years—and not from the contemporary production by either the land or ocean biosphere.
Fossil fuels and the air we breathe
How about future trends of atmospheric oxygen? As early as 1970, the prominent geochemist Wally S Broecker recognized that if we were to burn all known fossil fuel reserves, we would use up less than 3% of our oxygen reservoir.
If we were to cut or burn all forests and oxidize all organic carbon stored in vegetation and top soils worldwide, it would only lead to a small depletion in atmospheric oxygen. If photosynthesis in the ocean and on land stopped producing oxygen, we could continue breathing for millennia, though we would certainly have other problems.
The projected decline in atmospheric oxygen, even in the worst-case scenarios with massive fossil fuel burning and deforestation, will be very small relative to the very large atmospheric reservoir. Models show that the content of oxygen in the atmosphere will experience a minute change over the next 100,000 years in response to fossil fuel use. So while there are many things to worry about in our climate future, the availability of oxygen for air-breathing organisms (including humans) isn't one of them.
Oxygen decline in the ocean
There are significant causes for concern regarding the content of oxygen in the ocean, however. The ocean's O2 reservoir is vulnerable because it holds less than 1% of the oxygen stored in the atmosphere. In particular, ocean regions with very low or absent oxygen, referred to as oxygen minimum zones, expand as the planet warms, making new regions inhabitable for breathing organisms like fish.
The open ocean lost 0.5 to 3.3% of its oxygen stock in the top 1000 meters from 1970-2010, and the volume of oxygen minimum zones has increased by 3-8%.
This oxygen loss is primarily due to increasing ocean stratification. In this process, the mixing of the surface ocean, which becomes warmer and lighter, with the deeper and denser ocean layers is less efficient, restricting the penetration of oxygen. The activity of enzymes, including those involved in respiration, also generally increases with temperature. So, oxygen consumption by ocean creatures increases as the ocean warms.
A recent study found that oxygen minimum zones in the open ocean have expanded by several million square kilometers and hundreds of coastal sites now have oxygen concentrations low enough to limit animal populations and alter the cycling of important nutrients. The volume of low-oxygen areas is projected to grow by about 7% by 2100 under a scenario of high-CO2 emissions.
Deoxygenation of this kind affects biodiversity and food webs; and negatively affects food security and livelihoods of the people who depend on it.
The facts
So where does this leave our mantra?
While it is incorrect to say that the ocean provides 50% of the oxygen we breathe, it is correct to say that, over geological time scales, the ocean has provided a large fraction of the oxygen we take in today. It is also perfectly correct to say that the ocean is responsible for 50% of primary production on Earth, sustaining our food system.
And while we should not worry about the future supply of oxygen for humans to breathe in the future, we should worry about fish being increasingly displaced from expanding ocean areas that are depleted in oxygen.
Provided by The Conversation
This article is republished from The Conversation under a Creative Commons license. Read the original article.![]()




















