Highly efficient chainmail catalysts developed for decoupled water electrolysis
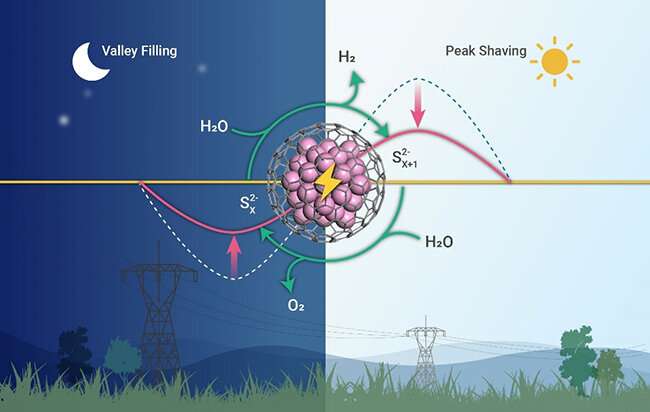
Decoupled electrolysis of water is a promising strategy for peak load regulation of electricity. It can store surplus electricity in the valley time and produce hydrogen with low energy consumption in the peak time, relieving the imbalance between the generation and consumption of electricity.
Recently, Prof. Deng Dehui's group from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) designed an efficient decoupled water device by using a kind of high-performance chainmail catalyst as the electrode.
Their study was published in The Innovation on July 14.
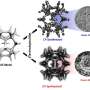
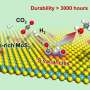
The researchers used the graphene-encapsulated CoNi chainmail catalyst as the electrode and polysulfides as mediators to develop decoupled water device.
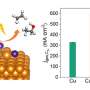
The device produced hydrogen with a low potential of 0.82 V at 100 mA/cm2. Compared with the direct electrolysis of water, the potential of hydrogen production was reduced by 1.24 V, saving 60.2% energy in the peak time.
With the efficient catalysis of chainmail catalyst, this device ran stably for 500 h at the current density of 500 mA/cm2, which reached high capacity of 2.5*105 mAh/cm2 for producing hydrogen.
Prof. Guan Jing's group from Qingdao University of Technology studied the reaction active by DFT calculations. They revealed that the superiority of chainmail catalyst originated from the modulation of the electronic structure of graphene surface by metal core and nitrogen dopant.
"This work provides a new route to the rational use of electricity in a separate period, which is helpful to construct smart power grids in the practical applications," said Prof. Deng.
More information: Mo Zhang et al, Highly efficient conversion of surplus electricity to hydrogen energy via polysulfides redox, The Innovation (2021). DOI: 10.1016/j.xinn.2021.100144
Provided by Chinese Academy of Sciences