Tiny, injectable sensors could monitor brain activity without surgery or implants
Researchers have developed nanoscale sensors that could be injected into the body to noninvasively track brain activity using light. The approach could one day offer a new way to study the brain or assess patients' brain functioning without the need for surgery or implanted devices.
A. Ali Yanik from the University of California, Santa Cruz, will report on the technology, called NeuroSWARM3, at the virtual OSA Imaging and Applied Optics Congress held 19-23 July. Yanik's presentation is scheduled for Tuesday.
"NeuroSWARM3 can convert the signals that accompany thoughts to remotely measurable signals for high precision brain-machine interfacing," said Yanik. "It will enable people suffering from physical disabilities to effectively interact with the external world and control wearable exoskeleton technology to overcome limitations of the body. It could also pick up early signatures of neural diseases."
The approach offers a new way to monitor electrical activity in the brain using a system-on-nanoparticle probe that is comparable in size to a viral particle. Neurons use electrical signals to convey information to each other, making these signals crucial to thought, memory and movement. While there are many established methods for tracking the brain's electrical activity, most require surgery or implanted devices to penetrate the skull and interface directly with neurons.
The researchers named their new technology Neurophotonic Solution-dispersible Wireless Activity Reporters for Massively Multiplexed Measurements, or NeuroSWARM3.
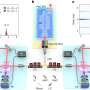
The approach involves introducing engineered electro-plasmonic nanoparticles into the brain that convert electrical signals into optical signals, allowing brain activity to be tracked with an optical detector from outside the body.
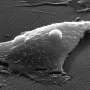
The nanoparticles consist of a silicon oxide core measuring 63 nanometers across with a thin layer of electrochromically loaded poly (3, 4-ethylenedioxythiophene) and a gold coating 5 nanometers thick. Because their coating allows them to cross the blood-brain barrier, they could be injected into the bloodstream or directly into the cerebrospinal fluid.
Once in the brain, the nanosensors are highly sensitive to local changes in the electric field. In laboratory tests, in vitro prototypes of the NeuroSWARM3 were able to generate a signal-to-noise ratio of over 1,000, a sensitivity level that is suitable for detecting the electrical signal generated when a single neuron fires.
"We pioneered the use of electrochromic polymers (e.g., PEDOT:PSS), for optical (wireless) detection of electrophysiological signals," Yanik added. "Electrochromic materials with optical properties that can be reversibly modulated by an external field are conventionally used for smart glass/mirror applications."
NeuroSWARM3 can be thought of as a nanoscale electrochromically loaded plasmonic antenna operated in reverse: instead of applying a known voltage, its optical properties are modulated by the spiking electrogenic cells within its vicinity. Hence, NeuroSWARM3 provides a far-field bioelectric signal detection capability in a single nanoparticle device that packs wireless powering, electrophysiological signal detection and data broadcasting capabilities into nanoscale dimensions.
The optical signals generated by NeuroSWARM3 particles can be detected from outside the brain using near-infrared light with wavelengths between 1,000-1,700 nm. The nanoparticles can function indefinitely without requiring a power source or wires.
Other researchers have explored a similar approach using quantum dots designed to respond to electrical fields. Comparing the two technologies, the researchers found NeuroSWARM3 generates an optical signal that is four orders of magnitude larger. Quantum dots required ten times higher light intensity and one hundred times more probes to generate a comparable signal.
"We are just at the beginning stages of this novel technology, but I think we have a good foundation to build on," said Yanik. "Our next goal is to start experiments in animals."
In addition to Yanik, the co-authors of this study include UCSC graduate students Neil Hardy, Ahsan Habib, and undergraduate researcher Tanya Ivanov.
More information: www.osa.org/en-us/meetings/osa … ied_optics_congress/
Provided by The Optical Society