Scientists discover faster way to manufacture vascular materials

Developing self-healing materials is nothing new for Nancy Sottos, lead of the Autonomous Materials Systems Group at the Beckman Institute for Advanced Science and Technology at the University of Illinois Urbana-Champaign.
Drawing inspiration from biological circulatory systems—such as blood vessels or the leaves on a tree—University of Illinois Urbana-Champaign researchers have worked on developing vascularized structural composites for more than a decade, creating materials that are lightweight and able to self-heal and self-cool.
But now, a team of Beckman researchers led by Sottos and Mayank Garg, postdoctoral research associate and lead author of the newly published Nature Communications paper, "Rapid Synchronized Fabrication of Vascularized Thermosets and Composites," have shortened a two-day manufacturing process to approximately two minutes by harnessing frontal polymerization of readily available resins.
"For the past several years we've been looking for ways to make vascular networks in high-performance materials," said Sottos, who is also Swanlund Endowed Chair and head of the Department of Materials Science and Engineering at Illinois. "This is a real breakthrough for making vascular networks in structural materials in a way that saves a lot of time and saves a lot of energy."
Garg said the simplest way to understand their work is to picture the composition of a leaf with its internal channels and structural networks. Now, imagine that the leaf is made from a tough structural material; inside, fluid flows through different spouts and channels of its interconnected vasculature. In the case of the researchers' composites, the liquid is capable of a variety of functions, such as cooling or heating in response to extreme environments.
"We want to create these life-like structures, but we also want them to maintain their performance over substantially longer times compared to existing infrastructure by adopting an approach biology uses ubiquitously," Garg said. "Trees have networks for transporting nutrients and water from the ground against gravity and transporting synthesized food from the leaf to the rest of the tree. The fluids flow in both directions to regulate temperature, grow new material, and repair existing material over the entire lifecycle of the tree. We try to replicate these dynamic functions in a non-biological system."
However, creating these complex materials has historically been a long, daunting process for the Autonomous Materials Systems Group. In previous research on self-healing materials, researchers needed a hot oven, vacuum, and at least a day to create the composites. The lengthy manufacturing cycle involved curing the host material and subsequently burning or vaporizing a sacrificial template to leave behind hollow, vascular networks. Sottos said the latter process can take 24 hours. The more complicated the vascular network, the more difficult and time-consuming it is to remove.
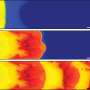
To create the host materials, scientists opt for frontal polymerization, a reaction-thermal diffusion system that uses the generation and diffusion of heat to promote two different chemical reactions concurrently. The heat is created internally during solidification of the host and surplus heat deconstructs an embedded template in tandem to manufacture the vascular material. This means the researchers are able to shorten the process by combining two steps into one, creating the vascular networks as well as the polymerized host material without an oven. Additionally, the new process enables researchers to have more control in the creation of the networks, meaning the materials could have increased complexity and function in the future.
"With this research, we've figured out how to put in vascular networks by using frontal polymerization to drive the vascularization," Sottos said. "It gets done in minutes now instead of days—and we don't have to put it in an oven."
Two processes in one: Tandem polymerization and vascularization allow scientists to create self-healing structural materials in a matter of minutes.
Self-healing materials can be beneficial wherever strong materials are essential to maintain function under sustained damage—such as the construction of a skyscraper. But in the case of the researchers, the most likely applications are for planes, spaceships, and even the International Space Station. Sottos explained materials produced in this manner could be commercially manufactured in five to 10 years, though the researchers note that all required materials and processing equipment are currently commercially available.
Beckman Institute Director Jeff Moore, a Stanley O. Ikenberry Endowed Chair of chemistry, as well as Philippe Geubelle, the Bliss Professor of aerospace engineering and executive associate dean of The Grainger College of Engineering, were also involved in the project.
From a computational standpoint, Geubelle explained that he was able to capture the frontal polymerization and endothermic phase change taking place in the sacrificial templates.
"We performed adaptive, transient, nonlinear finite element analyses to study this competition and determine the conditions under which this simultaneous frontal polymerization and vascularization of the gel can be achieved," he said. "This technology will lead to a more energy efficient and substantially faster way to create composites with complex microvascular networks."
Thanks to the team's interdisciplinary discovery, dynamic multifunctional materials are now easier to manufacture than ever before.
"This research is a combination of experimental work as well as computational work," Garg said. "It requires synchronized communication among team members from various disciplines—chemistry, engineering, and materials science—to overhaul traditional non-sustainable manufacturing strategies."
"There's nothing better than to see ideas bubble up from students and postdocs in the AMS group resulting from interactions and joint group meetings," Moore added. "The Moore Group has studied chain unzipping depolymerization reactions for years. I was delighted when I learned that the AMS team recognized how the thermal energy produced in a heat-evolving polymerization reaction could be synced to chain unzipping depolymerization in another material for the purpose of fabricating channels. The first time I saw Mayank's results, I thought to myself, 'I wish I'd have thought of that idea.'"
More information: Mayank Garg et al, Rapid synchronized fabrication of vascularized thermosets and composites, Nature Communications (2021). DOI: 10.1038/s41467-021-23054-7
Journal information: Nature Communications