December 21, 2020 feature
Designing high-performance hypergolic propellants for space rockets based on the materials genome
A new generation of rocket propellants for deep space exploration such as ionic liquid propellants with long endurance and high stability, are attracting significant attention. However, ionic liquid propellants are strongly restricted by their inadequate hypergolic (spontaneous ignition) reactivity between the fuel and the oxidant, where this defect can cause local burnout and accidental explosions during rocket launch. In a new report, Wen-Li Yuan and a research team in Chemistry at the Sichuan University in China and the Idaho University in the U.S. have proposed a visual model to demonstrate features of propellants to estimate their performance and applications. The materials genome and visualization model of the propellants greatly improved the efficiency and quality of developing performance propellants with applications to discover new and advanced functional molecules in the field of energetic materials. The work is now published in Science Advances.
Space exploration and the materials genome method.
A recent strategy to discover new materials presents a method based on "materials genomes," which relies on big data analysis of the structures and properties of target materials to discover new materials. Researchers aim to construct artificial intelligence programs and screening to analyze a large number of possible structures in a short timeframe using the method. Yuan et al. applied the materials genome method in this work to predict the most probable hypergolic additive. Humans have always been fascinated with space exploration and have envisioned traveling through space-time, although technological limits have withheld this ambition for thousands of years. At present, high-performance spacecraft are in development with modern technologies to achieve manned and unmanned space navigation within low earth orbit and across our solar system.
Chemical energy released by a propellent (rocket fuel) forms the power source of rockets and spacecraft, and can determine the altitude range and service life of a spacecraft. Examples include the Atlas-Centaur rocket based on liquid dihydrogen and oxygen fuel, bound for Mars and Venus, as well as the Long March 3B rocket containing UDMH (unsymmetric dimethyl hydrazine)/dinitrogen tetroxide to the moon. However, these high-performance rocket propellants or fuels are limited by high toxicity and decomposition, alongside their stable existence only at extremely low temperatures. The need for an efficient and systematic method to design high-performance hypergolic additives therefore exists. The materials genome method can reduce the period of investigation required to develop such new materials.
To establish a hypergolic materials genome database, the team identified key structures of hypergolic compounds and explored their structure-activity relationships. A hypergolic reaction is an exothermic redox reaction (i.e., combustion) where components can spontaneously ignite on contact in a rocket combustor. Such compounds are typically made of gas-generating elements such as carbon and nitrogen. Much like the relationship between the gene and its base pair, the diverse hydrogen (H), carbon (C), nitrogen (N) and other elements constitute a series of hypergolic functional groups and frameworks to generate hypergolic compounds as suitable rocket propellants. The materials had to have an ignition delay time, a high enthalpy of combustion and a high specific impulse to determine the total energy payload capacity of rockets. Propellent additives should also be stable and compatible. Based on these requirements, Yuan et al. provided a direct method to identify key structures of hypergolic additives from the elemental composition of their functional structures.
Nitrogen and carbon-rich energetic propellants
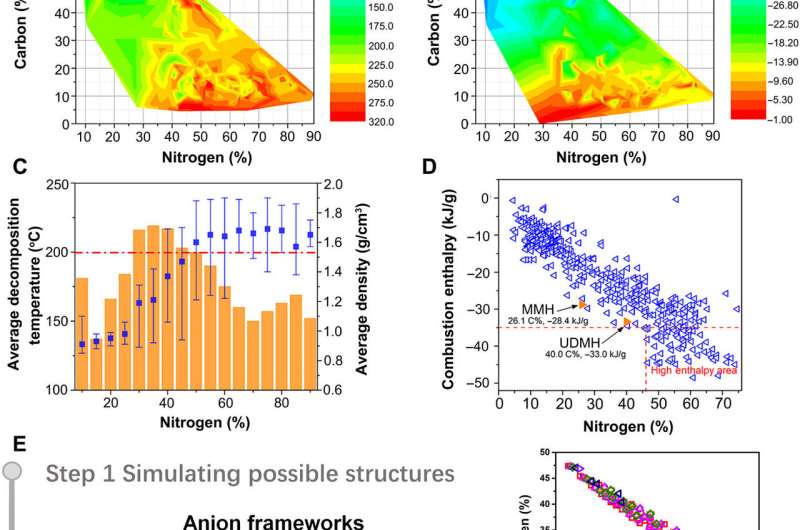
Nitrogen-rich energetic propellants can boost energy beyond traditional fuels to improve the specific impulse of rocket fuels. Using existing literature, the researchers found the relationship of more than 1000 propellants and their mixtures to understand the connection between their elemental composition and thermal decomposition properties. Propellants containing 30 to 50 percent nitrogen content had the highest thermal stability with decomposition temperatures in excess of 200 degrees Celsius. The researchers deduced an appropriate nitrogen content to meet the specific requirements and thermal stability for high-performance propellants. The carbon element content also generated substantial amounts of combustion heat and gaseous carbon dioxide necessary for spacecraft propulsion to provide sufficient chemical energy to overcome gravity. Based on the enthalpy of combustion between carbon and nitrogen, the enthalpy of propellant combustion was positively related to the carbon content. To design the propellants, the team combined the limits of nitrogen elements in propellants with a highest allowable carbon content to achieve the best performance for specific impulse and combustion enthalpy.
Structural composition and quantum analysis
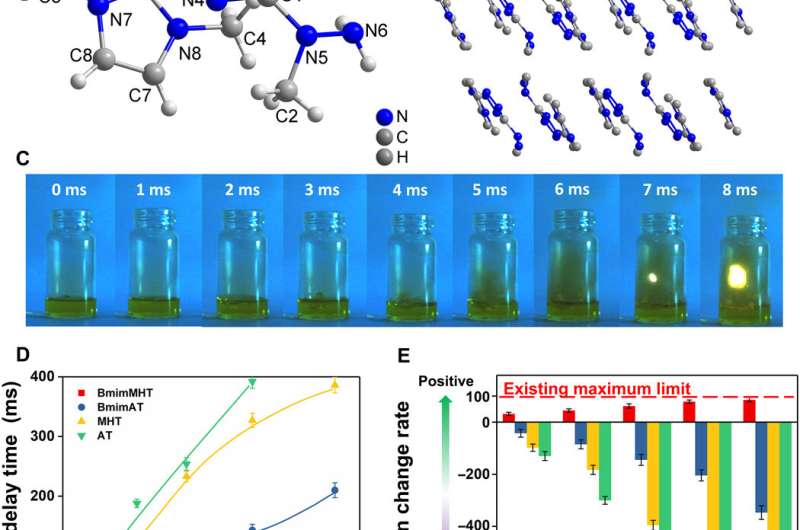
The structural composition was another key feature of high-performance propellants to determine their stability, ignition behavior and biological toxicity. Ionic liquids composed of cations and anions have unique advantages of miscibility, volatility, hypotoxicity and thermal stability to greatly reduce the risk of exposing the operator to aerosols and deflagration. Using a screening method, Yuan et al. provided basic guidance to rapidly design and identify target compounds and considered other important indicators, including hypergolic reactivity and density, to select the best performance structure.
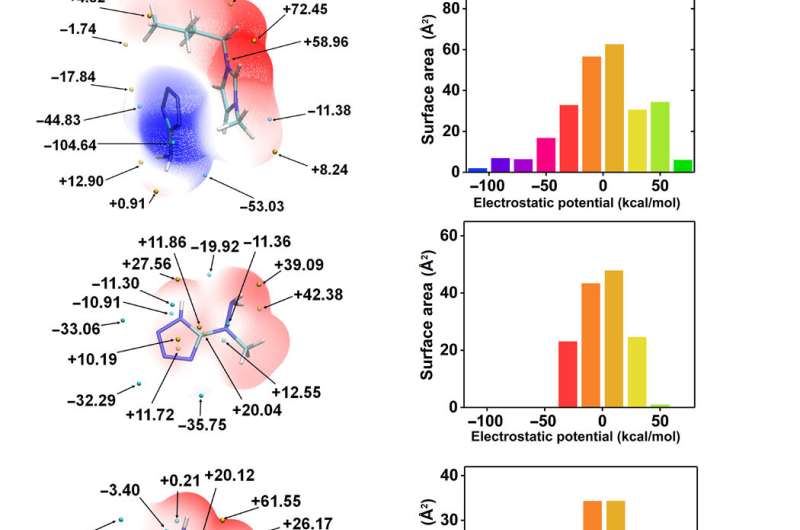
The team then conducted quantum analysis by investigating molecular orbital (MO) theory of anions as a criterion to determine hypergolicity and tested 15 anions, of which (1-methylhydrazinyl)tetrazolate (MHT) ionic liquids met all requirements of hypergolic additives. The genome database and screening process was therefore complete. Yuan et al. then studied the structure and physicochemical properties of MHT ionic liquids, including the density, thermal stability and detonation properties. Incidentally, the 1-butyl-3-methylimidazolium cation (Bmim+)-based MHT fuel had the highest thermal decomposition temperature, beyond 200 degrees Celsius, which was safe under extreme conditions in space. The team also tested two additional (Bmim+)-based propellants including Bmim-based dicyanamide (BmimDCA) and Bmim 5-aminotetrazole (BmimAT) ionic liquids.
Outlook—high-performance propellants
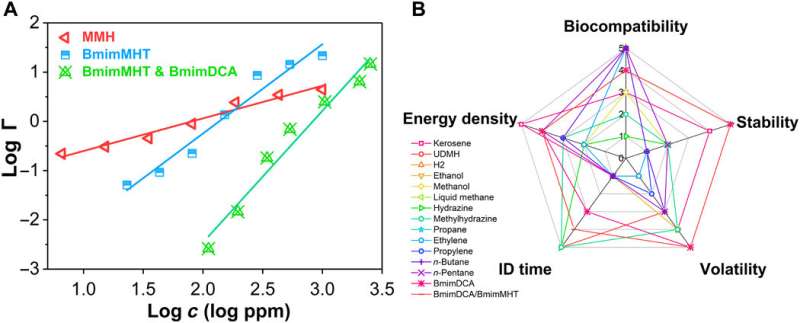
Since toxicity was a serious problem in propellants, the team tested the toxicities of the ionic liquids using a Vibrio fischeri bacterium that can determine the environmental acceptability and toxicology parameter of materials. The combined BmimMHT/BmimDCA ionic liquids were advantageous as green propellants compared to traditional fuels. The DCA ionic liquid was more unique relative to toxicity, stability and volatility. Based on the guidance of the material genome method of propellants, Yuan et al. combined the DCA ionic liquid with BmimMHT, to make up for the insufficient hypergolic behavior of DCA.
In this way, Wen-Li Yuan and colleagues designed a previously unrealized family of high-performance propellant by using the propellant materials genome method. The MHT ionic liquid successfully solved the ignition behavior of the DCA ionic liquids. The design strategy summarized the structure-activity relationship of propellants combined with stability, hypergolicity and toxicity in a first-in-study materials genome method integrated in the field of propellants. The genome approach will guide and promote the molecular design and application of new materials to develop new high-performance propellants.
More information: Yuan W. et al. Designing high-performance hypergolic propellants based on materials genome, Science Advances, 10.1126/sciadv.abb1899
Gibney E. First private Moon lander heralds new lunar space race, Nature, doi.org/10.1038/d41586-019-00634-8
Boyd P.G. et al. Computational development of the nanoporous materials genome, Nature Reviews Materials, doi.org/10.1038/natrevmats.2017.37
Journal information: Nature , Science Advances
© 2020 Science X Network