Depth-dependent valence stratification in a lithium-rich layered cathode
A team of scientists from the Beijing Synchrotron Radiation Facility at the Institute of High Energy Physics of the Chinese Academy of Sciences, the Stanford Synchrotron Radiation Light Source at SLAC and Brookhaven National Laboratory used synchrotron-based nano-resolution spectro-tomography to study a typical lithium-rich nickel-manganese-cobalt (LirNMC) material (i.e., Li1.2Ni0.13Mn0.54Co0.13O2) to visualize its multi-layer morphology and the chemical and spatial dependence of oxygen redox behavior.
This study, published in Nature Communications, reveals that the oxygen redox induces the depth-dependent transition metal valence stratification in LirNMC.
As a high-efficiency energy storage device, the Li-ion battery is widely used in electronic devices and electric vehicles. The research community has devoted great effort to improving electrochemical performance.
The LirNMC layered cathode is one of the promising candidates for the Li-ion battery because of its higher energy density. However, it suffers from voltage decay upon cycling. To overcome this problem, it is necessary to understand the mechanism for this voltage fade.
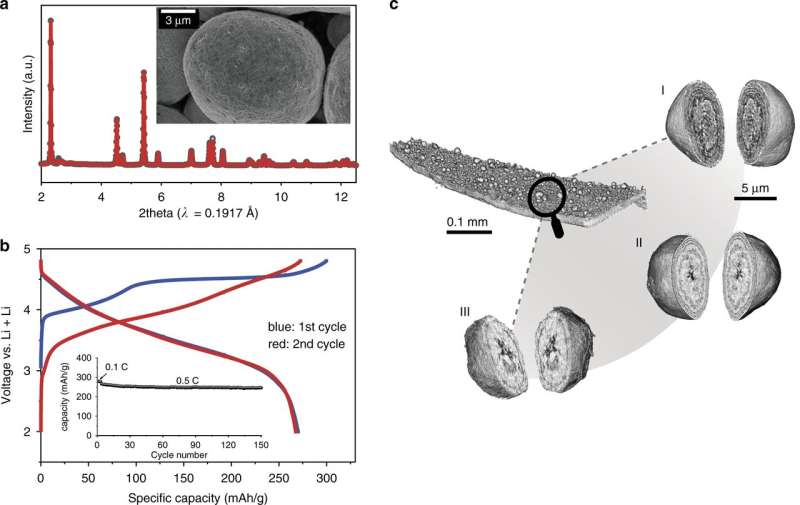
In this study, the team used nano-resolution spectro-tomography to study the LirNMC material at the particle level in three dimensions. This allowed visualization of the material's unique multi-layer morphology with homogeneous composition, and the charge-induced depth dependence of the transition metal valence distribution.
The soft X-ray resonant inelastic X-ray scattering (RIXS) and super partial fluorescence yield (sPFY) results clearly showed features of oxygen redox in the charged sample (4.8 V in the first cycle). Although oxygen activity can increase the capacity of the cathode, it will generate oxygen vacancy around the transition metal cations and decrease their valences. This is why the transition metal valence of LirNMC has a very different depth profile from that of conventional NM in the charged state.
This study highlights the importance of material engineering at the particle level and a depth-dependent compositional engineering strategy, which could be a viable way to solve the voltage fade problem in LirNMC cathodes.
More information: Jin Zhang et al. Depth-dependent valence stratification driven by oxygen redox in lithium-rich layered oxide, Nature Communications (2020). DOI: 10.1038/s41467-020-20198-w
Journal information: Nature Communications
Provided by Chinese Academy of Sciences