A new method for making cheaper chemicals
A new technique to make cheaper more efficient biological enzyme hybrids could have valuable applications in future water recycling, targeted drug manufacturing and other industries, Flinders University green chemistry researchers say in a new publication.
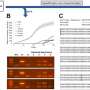
The model enzyme system, which immobilizes a catalyst enzyme hybrid for continuous flow use in the high-speed vortex fluidic device, showed a 16-fold increase in its efficiency, the researchers say in American Chemical Society journal, ASC Applied Materials & Interfaces.
Flinders Professor of Clean Technology Colin Raston, from the Flinders Institute for Nanoscale Science and Technology, with collaborator Professor Greg Weiss at University of California Irvine and other researchers around the world, have extensively used the vortex fluidic device in a wide range of applications—many of which promise to open new frontiers in clean manufacturing and even new industries.
Lead author on the new paper, Flinders University research associate Dr. Xuan Luo, says the cost and limited life of enzymes hinders development of enzyme-based biosensors and most enzymes are made inactive during the assay process so cannot be separated for reuse.
"We used an inorganic composite to trap the enzyme on the surface of the vortex fluidic device, essentially making a 'mini factory' where the enzyme was reused under continuous flow," Dr. Luo says.
"The technique uses the minimum amount of enzyme, which is less expensive, and monitors the reaction in real-time, also saving time and money on reagents."
Professor Raston, a finalist for South Australian Scientist of the Year 2020, says the paper demonstrates four applications of the vortex fluidic device—fabrication, immobilization, continuous flow and real-time monitoring.
"In this study, we were able to generate and immobilize laccase nanoflowers into silica hydrogel to greatly simplify the fabrication process, and allow saving of both time and money, along with the ability to reuse the enzyme for further reactions," says co-author Professor Raston says.
"The next steps will be to test the model system with actual samples such as wastewater, and also use this same immobilization system with other enzymes to see if their efficiency is increased."
The paper describes the immobilization of hybrid protein-Cu3(PO4)2 nanoflowers to create a new laccase nanoflower immobilization platform, LNF@silica, which subsequently increased the enzyme efficiency by 16 times and allowed assay monitoring in real-time.
More information: Xuan Luo et al, Vortex Fluidic-Mediated Fabrication of Fast Gelated Silica Hydrogels with Embedded Laccase Nanoflowers for Real-Time Biosensing under Flow, ACS Applied Materials & Interfaces (2020). DOI: 10.1021/acsami.0c15669
Journal information: ACS Applied Materials and Interfaces
Provided by Flinders University